New Drug Formulations: Loop Diuretics
It’s ok, unicorns. I, too, know that feeling of missing the boat…
Steph’s Note: Every once in a while, you run across a news article that makes you go, huh, that’s cool…why didn’t I think of that? Or at least that was my thought when I saw the articles about the approval of 2 novel diuretic formulations for use at home. (Could’ve been rich…sigh. I guess I’ll keep working lol).
So that we can all learn more about how old drugs can learn new tricks, Jenny Hoang grabbed hold of this topic baton and fleshed out what you need to know about these cool new drug delivery systems. (If you haven’t read anything by Jenny yet, you should - herbals, CAR-T, and PISCES.) Thanks again, Jenny, take it away!
Just about any pharmacist has seen a good ol’ case of edema, or fluid retention, in their career. You may recall that it is common in patients who have heart failure or chronic kidney disease. And you (hopefully) remember that we often use diuretics to alleviate this swelling. This is because these medications work in the kidneys to adjust the balance of ions and water so that the body doesn’t hold onto all the excess water causing the edema.
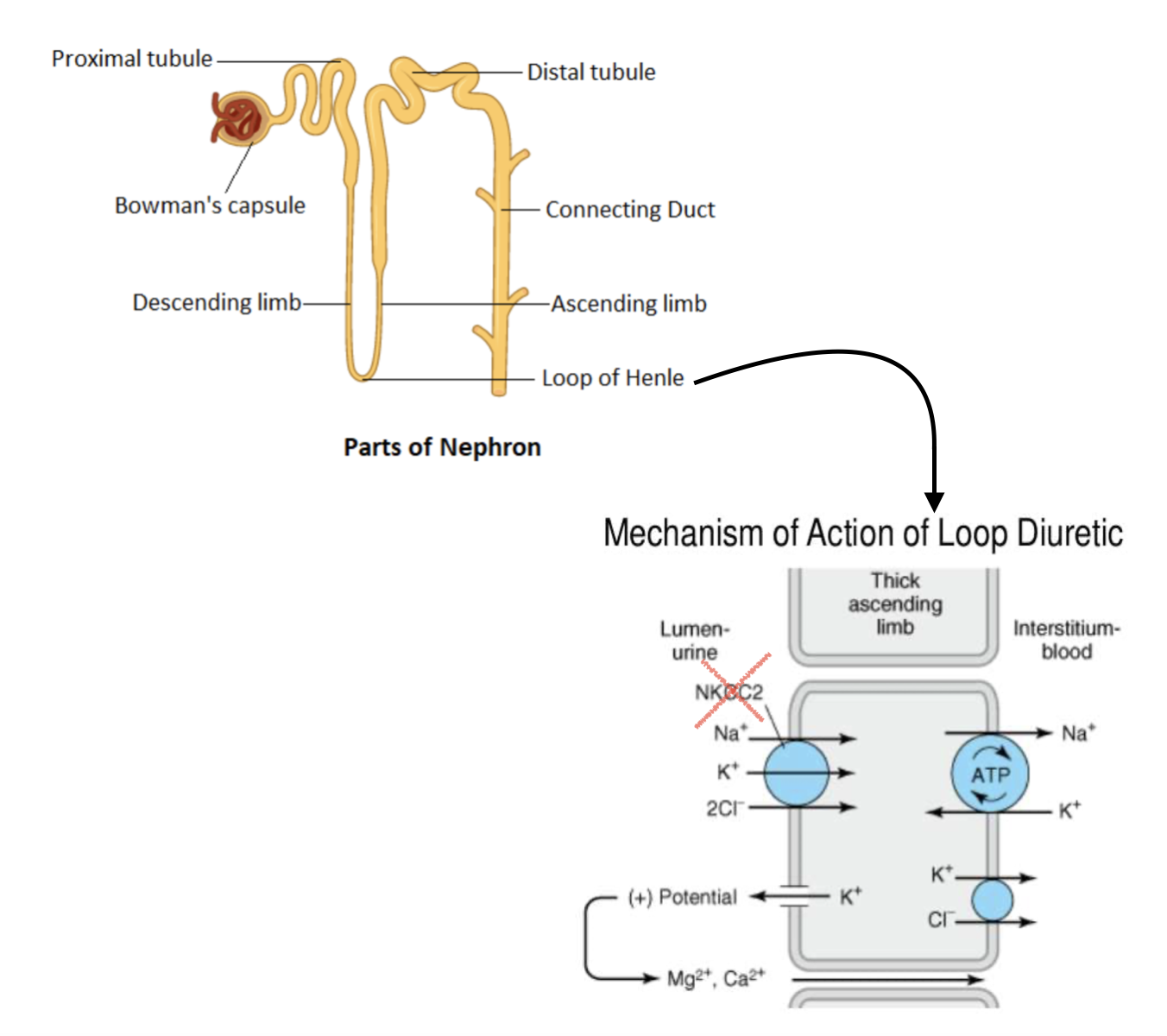
Specifically, we tend to see loop diuretics. They are the first line of treatment for congestion in heart failure patients. Loop diuretics work when the kidney is filtering the blood and is sorting out how much water and salt to keep. If we look at the nephrons that are inside the kidney, loop diuretics act at the Loop of Henle.
Loop diuretics block the NKCC2 symporter that transports Na/K/2Cl. Basically, keep the salt in the urine, water will follow.
Normally at the Loop of Henle, a lot of salt is absorbed. And remember that water follows salt. So if the Loop of Henle reabsorbs a lot of salt, then it makes sense that a lot of water is also reabsorbed. (Image)
But in the case of edema, we do not want more water to be absorbed, so we need to stop this. Loop diuretics come to the rescue by blocking salt absorption at the Loop of Henle. By doing this, salt stays in the urine, drawing water out with it.
The result? Less edema as less fluid is retained.
Pharmacokinetics of Loop Diuretics
Now let’s think about some real life application of loop diuretics. They are available in both IV and PO forms. The most common loop diuretic is furosemide.
Oral furosemide takes anywhere from 30 minutes to an hour to start working, and its action lasts about 6 hours (hence why the brand name is Lasix…get it, LaSIX). Oral tablets are highly effective for maintenance diuretic treatment. Patients can be monitored through routine check ups and can also watch their weight to have a good idea of how much fluid they are retaining, which allows them to adjust their dosing at home.
On the other hand, the IV version takes only minutes to start working, but more is excreted via urine compared to the oral tablet. So it only lasts about 2 hours.
Check out this handy dandy chart of loop diuretic doses.
We should also consider the dosing differences between IV and PO furosemide. Furosemide’s bioavailability is about 50%. This means that, when given orally, about 50% of the dose is lost to metabolism. This means only the other 50% that is absorbed is actively blocking salt retention.
So let’s look at a quick example. You have a patient who was hospitalized and stabilized on a dose of furosemide 20 mg IV daily, and he is now ready for discharge. What should his outpatient furosemide dose be? His IV dose should be doubled to 40 mg when converting to oral tablets for discharge.
One of the biggest problems however with the oral tablet is that if a patient has severe edema, they may have a lot of fluid stuck in their gut. This can cause highly variable absorption of the oral tablet, which can cause it to be not as effective. Due to this, despite the accessibility of oral tablets, patients may find themselves having to come to the hospital for IV loop diuretics.
Intravenous diuretics come in handy for situations such as acute pulmonary edema, heart failure, impaired gut absorption, intubation, or severe nausea and vomiting. The IV formulation is also a lot more predictable since it goes directly into the bloodstream. And another big plus about IV diuretics is that they allow for a quick, high plasma concentration that passes the threshold to be effective.
Many patients who chronically use loop diuretics may find that they have blunted responses due to the kidneys compensating through different mechanisms. (Because of this, some patients have to take another type of diuretic such as a thiazide diuretic). Some patients have blunted diuretic responses because they are so overloaded with fluids that they are not effectively absorbing higher levels of diuretics. (These patients have to keep taking higher and more frequent doses to get the same effect).
While IV administration helps eliminate those issues, the drawback to IV diuretics is that they require hospitalization, which is costly and time consuming - not to mention increases the burden on the healthcare system. There are also those patients who simply don’t recognize the severity of their condition soon enough and fail to get to the hospital in time for help.
If only there were solutions at home…
A New Subcutaneous Loop Diuretic Formulation
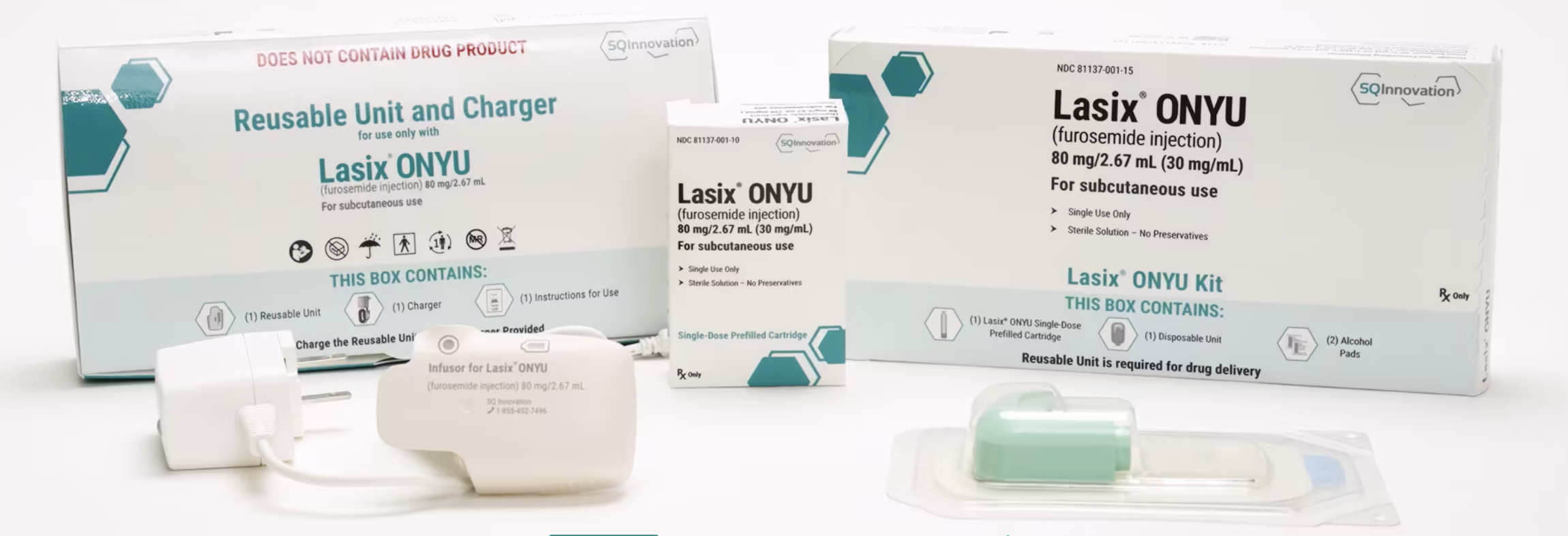
Home is where my bathroom is. This is the proud slogan of SQ Innovation INC’s new drug Lasix ONYU. It is a wearable (ONYU, “on you,” wink wink) drug delivery device that releases 80 mg of furosemide in 2.67ml prefilled syringes subcutaneously through an infuser. Each infusion lasts about 5 hours and can be used once or twice a day, with typical treatment lengths from 3-10 days. Patients can expect to go to the bathroom a lot during that time.
The infuser consists of a rechargeable, reusable unit that attaches to a disposable unit that is attached to the skin (stomach area, rotating sites each use). It releases the prefilled syringe with the medication dose. Each reusable unit can be used for 48 treatments. And best of all, patients can easily use it at home without needing to go to the doctor or hospital! (Image)
Everything a patient would need to get started with SC furosemide.
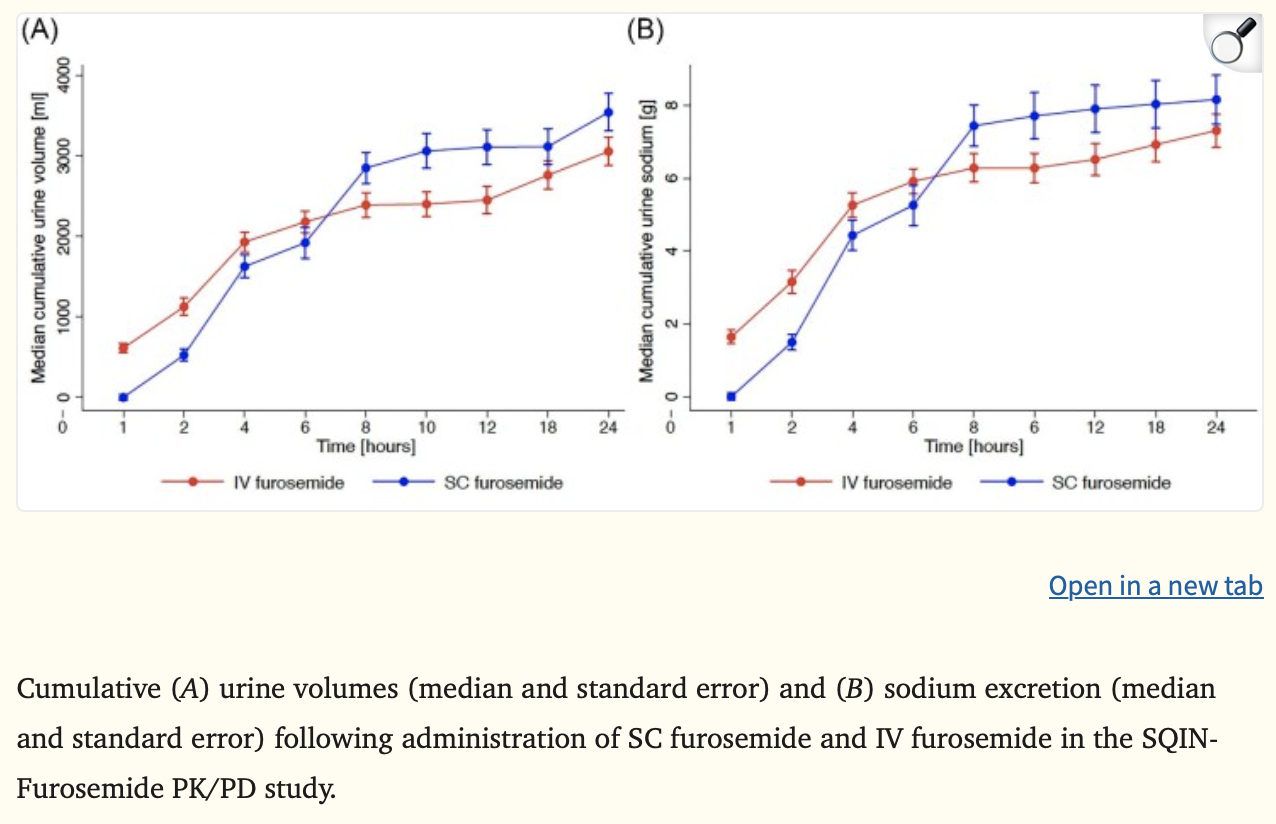
In the 2023 SQIN-Furosemide PK/PD trial, they tested 80 mg intravenous (IV) furosemide head to head with the 80 mg subcutaneous (SC) injectable furosemide in ~2.7ml with the primary outcome of absolute bioavailability of the SC furosemide. The resulting bioavailability was found to be 112% for SC compared to IV. The urine volume percent in 8 hours was 116% with 118% sodium excretion, and 115% urine volume in 24 hours for SC vs. IV. (Image)
This tells us that the SC formulation performed just as well as, if not slightly better than, the IV formulation, especially with increasing time from dose. Few patients reported transient skin irritation with the SC injection and most patients found the application very usable and acceptable for their use. Overall, this study demonstrated that the SC formulation was very promising.
However, it should be noted that this first study was conducted in a small group of 18 participants and all were in stable heart failure using oral diuretics. So, what about clinical outcomes instead of PK/PD? And what about in decompensated patients rather than stable?
Enter the 2024 AT HOME-HF study, conducted by scPharmaceuticals on their subcutaneous furosemide on-body device FUROSCIX. In this study, 51 outpatients with chronic HF and increasing fluid congestion were randomized 2:1 to either SC furosemide or usual care. The primary outcome was a win ratio of a 30 day composite of cardiovascular death, HF events, and changes in N-terminal pro-BNP levels (a marker of fluid overload). Secondary endpoints were related to patient functionality and quality of life.
Side bar - what’s a win ratio? Glad you asked because I had to look it up too!
A win ratio is a statistical representation of wins versus losses (aka good outcomes in the treatment group vs bad outcomes in the control group). A win ratio of greater than 1 means the treatment helped. It’s different than other outcome statistics in that it accounts for the order in which patients and outcomes are compared, allowing for some prioritization of outcomes. That is, patients may be compared according to hierarchal levels. First may be death rates (as that is the most important consequence!), but if that’s equal between groups, then the comparison moves on to the next component of the endpoint (e.g., HF events in the case of the AT HOME-HF study). Pretty interesting representation of outcomes when we’re so used to seeing time-to-event hazard ratios in most studies.
Alright, now that we’ve had our little statistics side bar, back to the study results for SC furosemide. The SC furosemide to usual care win ratio was 1.11 (95% CI 0.48-2.50, p=0.806). Bit difficult to interpret with that wide confidence interval and nonsignificant p-value…not super great there. Of course, there were only 51 people in the study total, so perhaps a missed significance due to insufficient sample? IDK, TBD. The positive news was that the SC furosemide group did experience a significantly greater reduction in weight at day 3 than the usual care group, as well as improved dyspnea and functionality scores. So perhaps there’s something there…further studies needed!
A New Intranasal Loop Diuretic Formulation
Let’s take a look into a new nasal spray form of bumetanide, which is another loop diuretic. As we’ve already discussed, IV is often needed for quick and effective diuresis in emergency situations, but it can prove to be inconvenient and costly. The new Bumetanide Nasal Spray (BNS, brand name Enbumyst) is designed to provide an intranasal solution that is both quick and highly bioavailable. (Image)
As we discussed earlier, oftentimes those characteristics are difficult to achieve at home with oral diuretics, which is why people require IV diuresis. Additionally, if patients have excess fluid retention, they may have gut edema that will hinder absorption of their oral maintenance doses. So this is where the nasal spray can be a great solution as it bypasses those absorption issues.
The BNS is designed to be highly absorbed through the nasal mucosa. In this randomized PK/PD open label crossover trial of the BNS, they looked at the primary outcomes of Cmax and AUC for BNS versus oral and IV bumetanide. They found that BNS was bioequivalent to oral bumetanide, and there were no differences in Cmax and AUC between the two as well. However, the BNS did reach max concentrations (Tmax) more quickly than the oral tablet (1 hour versus 1.5 hours, respectively). There were also no differences in urine output at 2 and 24 hours or in sodium excretion.
The researchers also noted that both the BNS and IV formulations only had 27% variability in absorption instead of the over 40% for the oral formulation. Patients found the formulation comparatively tolerable to other formulations, and no overt nasal irritation occured. But again, they recruited healthy individuals, who may not be representative of those needing diuresis the most.
While both the new SC formulation and this BNS provide excellent diuretic options at home, the BNS is less invasive and simpler to use. Another consideration is that, if a patient has nasal congestion, they may not experience the same absorption observed in the studies, so the SC option may be preferred in that instance.
The tl;dr of New Loop Diuretic Formulations
All in all, loop diuretics are very effective at reducing fluid retention. In many disease states, such as heart failure or chronic kidney disease, they are often used orally for fluid maintenance. Oral formulations, however, can be finicky when it comes to gut absorption, especially in patients with lots of edema. Increasingly higher doses may be required to achieve the same effect.
In emergency situations, such as acutely decompensated heart failure, patients may need to be hospitalized to receive IV loop diuretics for better bioavailability and quicker, more effective fluid reduction. This is time consuming and costly, so drug manufacturers are creating alternatives to at-home oral doses to provide the efficacy of IV formulations without the need to be hospitalized.
There are a lot of promising data for both a new SC and nasal formulation of loop diuretics. In small, preliminary studies, these new formulations were found to be similar to IV in kinetics and effects. As more studies are conducted, especially in patients who more severely decompensated, we will find out if these new formulations actually live up to their promise of IV efficacy in the comfort of home.