Where Have All the Kids' Cold Meds Gone?
Steph’s Note: As a mom of 3 kids 4 years old and younger, I’m currently in a phase of life that involves bouts of health. You read that right. Not bouts of illness…bouts of health. They say we’re building our immune libraries, but honestly, it just seems like we’re succumbing to snot and croup at every turn. (For those of you who aren’t parents who just cringed that I used the word “snot”…just wait. There’s more.)
As a pharmacist mom stuck in the seven levels of phlegmy hades, I have had soooo many instances of wishing there was more I could do for my sweet barking babies. I mean, when I was a kid, we had Robitussin (the yucky red one) and Dimetapp (the yummy purple one, IYKYK). So what happened to these options? Why are they now only recommended for older kids?
In order to answer these questions that moms (and grandmas) everywhere are asking, we’re going to turn the tables a bit today. Rather than discussing what we DO use for treatment, we’re going to talk about the medications we DON’T use in kids - and why. Let’s go.
Blacklisted Medication #1: Dextromethorphan
According to the FDA and the American Academy of Pediatrics (AAP), dextromethorphan should not be used for cough suppression in children under the age of 4 years. (UK guidelines are even more restrictive with a minimum age of 6 years.) And even between ages 4-6 years, it’s not recommended first line and should only be used under the advisement of a medical provider. Instead, they recommend honey as a first line intervention (assuming the child is over the age of 1 year given the risk of botulism).
Now, for all my fellow parents out there, have you tried honey for your child’s cough? Did it do a darn thing?
Yeah. Me neither. Batting 0 on that one. (Image)
So alas, what’s the deal with dextromethorphan. What is this medication, and why isn’t it on a list of available cough suppressants for young kids?
Dextromethorphan is a centrally acting antitussive. While its exact mechanism is still unknown, it’s thought to relieve coughing through a combination of the following:
Action in the nucleus tractus solitarius, which is a part of the brainstem involved in the cough reflex
Sigma-1 receptor agonist, which is involved in neuronal excitability and the cough reflex
Of note, dextromethorphan is a bit of a jack of all trades. While the above are thought to be targets for relieving coughing, it has many other targets that make it useful for treatment of other conditions as well. For example, it is a noncompetitive antagonist at NMDA receptors and also inhibits serotonin and norepinephrine transporters, which contribute to its antidepressant-like effects. It also blocks several nicotinic acetylcholine receptors and some voltage-gated calcium channels. It’s approved for pseudobulbar affect (PBA) in combination with quinidine and major depressive disorder in combination with bupropion, and it’s being studied for everything from stroke to autism.
Its NMDA antagonism at abusive doses is also what produces the “robo-tripping” high, which is why many states have enacted legislation to restrict purchase to people over the age of 18 years. For context, NMDA antagonism is how ketamine and PCP create euphoria, dissociation, and hallucinations.
Who knew it was all in a bottle in every OTC cough and cold section across the country. Isn’t it wild sometimes to think about what’s sitting on the shelves available for purchase?!
A moment about what dextromethorphan is NOT (because I used to be under this misconception too!). Dextromethorphan is NOT an opioid. It does not act on the mu, kappa, or delta opioid receptors. It is also not a codeine derivative. So when you think dextromethorphan, do not think opioid.
I think some of my own confusion surrounding this dextromethorphan-codeine situation came from its metabolism. Like codeine, which requires metabolism by CYP2D6 to produce its active metabolite (morphine), dextromethorphan also requires metabolism by CYP2D6 to produce its active metabolite (dextrorphan).
Much like the issues surrounding pediatric use of opioids, especially codeine, when CYP2D6 metabolizer phenotype is unknown (aka pretty much everybody), not every child metabolizes dextromethorphan the same way. This variability can lead to a wide spectrum of efficacy and safety. Poor metabolizers won’t make enough active metabolite to be effective, whereas extensive/rapid metabolizers risk toxicity and overdose.
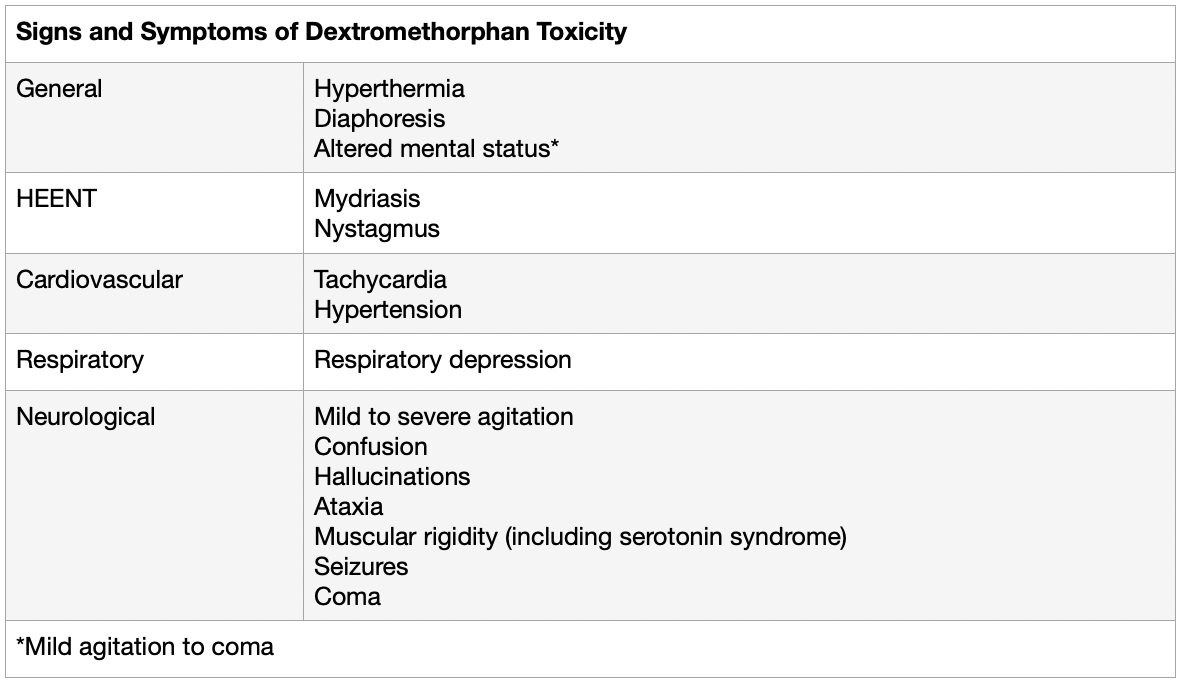
Speaking of overdose, what does this look like for dextromethorphan?
At this point, we’ve pretty well established that this OTC medication has a few skeletons in the closet. But what drug doesn’t? Nothing is completely safe. We give acetaminophen and even ibuprofen to infants (albeit greater than 6 months for ibuprofen), and those meds certainly have some baggage too. So what’s the nail in the coffin for dextromethorphan in kids, if it’s not just safety issues?
The other side of the coin: effectiveness. There haven’t been any studies supporting dextromethorphan efficacy or superiority in young children. In an oft cited JAMA study from 2007, a single bedtime dose of dextromethorphan was compared with honey and no treatment in 105 children 2-18 years old (mean age 5.22 years). Parents were surveyed in a before and after fashion about cough severity, cough frequency, how bothersome the cough was, and sleep quality for child and parents. Honey produced the greatest improvements in symptom scores overall. When compared with the other 2 arms, honey was superior to no treatment, and there were no significant differences between honey and dextromethorphan.
So basically, why use dextromethorphan in kids when there’s a safer alternative that appears to be just as good, if not better?
(Again, not trying to be a Debbie Downer here, but from personal experience, if honey is the best we have, can we get hopping on developing something better? Please? But I digress. Might have some cough-induced sleep deprivation trauma at the moment from bed hopping with sick kids.)
Alright, I think that gives you the need to know on dextromethorphan. The tl;dr? Potential safety issues surrounding metabolism and toxicity combined with a lack of proven efficacy make this a no go for young coughing kids.
Blacklisted Medication #2: Brompheniramine
I titled this section brompheniramine, but in reality, this section is really about first generation antihistamines, including brompheniramine, diphenhydramine, chlorpheniramine, and doxylamine. The FDA and AAP warn of significant danger in children under the age of 2 years, and they recommend against use in kids under 6 years due to concerns surrounding lack of efficacy and safety. Let’s take a closer look.
First generation antihistamines are highly lipophilic, meaning they are able to easily cross the blood brain barrier. They antagonize both peripheral and central histamine-1 (H1) receptors, allowing them to ameliorate histamine-mediated IgE and non-IgE allergic conditions. However, they also produce (often undesirable) CNS effects, including sedation, as well as anticholinergic effects, including the following lovely rhyme:
Can’t see: blurry vision
Can’t spit: dry mouth
Can’t pee: urinary retention
Can’t *poo emoji*: constipation
So while it may be tempting to use these first generation antihistamines for their anticholinergic and CNS side effects to dry up snotty noses and sedate a poor sick kid into sleeping at night, there’s more to the story.
In this 2024 JAMA study of more than 3000 children in Korea, use of first generation antihistamines was associated with a 22% increased seizure risk (adjusted OR [AOR], 1.22 [95% CI, 1.13-1.31]). That risk was even more pronounced at 49% in children aged 6 to 24 months (AOR, 1.49 [95% CI, 1.31-1.70]). Risk in children aged 25 months to 6 years was less clear (AOR, 1.11 [95% CI, 1.00-1.24]; P = .04 for interaction).
Of note, the first generation antihistamines included in this study were chlorpheniramine maleate, mequitazine, oxatomide, piprinhydrinate (actually a combination of an H1 antagonist and a xanthine stimulant like theophylline), and hydroxyzine hydrochloride. Not all the same products as what’s available here in the US, but some are.
So strike #1 - increased seizure risk, especially in children under 2 years. (Image)
In this 2023 study of almost 2000 Korean children, use of first generation antihistamines was associated with ~20% increased risk of cardiovascular events. Even when they excluded patients who had a known cardiac condition or history, there was STILL an observed ~20% increased risk! So the risk was elevated independent of baseline cardiac status. While ischemia was observed, cardiac arrhythmias were noted to be the most prominent issue (over 95% of events).
This isn’t too awfully surprising when you think about it. Consider the warnings of QT prolongation and Torsades de Pointes (TdP) on hydroxyzine and promethazine, which are other first generation antihistamines. They’re just sharing the cardiac love with their rhinorrhea-treating cousins.
So strike #2 - increased risk of cardio toxicity, especially arrhythmias.
Then we come to the third strike, which is altered mental status. For most users of first generation antihistamines, sedation is the notable mental effect. However, in pediatrics especially, there is a subset of patients who experience paradoxical excitation and/or hyperactivity. (My mom tells the story of how my toddler brother banged his head repeatedly on the floor after receiving diphenhydramine in the 80s…no bueno.)
Beyond the risk of adverse events at therapeutic doses, there is also the risk associated with accidental or intentional overdoses when trying to get sick kids to sleep. In overdose situations, first generation antihistamines can contribute to respiratory depression - which can be fatal. According to one ABC News report from 2008, when many children’s cough and cold products were pulled from the shelves and/or relabeled, there were 69 reports of pediatric fatalities in the setting of first generation antihistamines between 1969 and 2006.
So there you have it, strike #3 - increased risk of altered mental status, both over sedation/respiratory depression and hyperactivity.
Rather than reaching for these first generation antihistamines that don’t have great efficacy data and certainly have some safety baggage, most clinicians prefer to recommend second generation antihistamines - such as cetirizine - for assistance with red, runny noses and congested chests. These second gens have longer durations of action and better safety profiles due to their selectivity for peripheral H1 receptors (rather than central). Cetirizine is approved for children 6 months and up.
Blacklisted Medication #3: Vick’s VapoRub
I know, I know…yes, I’m serious. The third blacklisted product isn’t even a “medication”? How bad could it be that it’s on the blacklist as a topical natural product?!?
Let me pull out my soapbox one more time. Natural products aren’t any safer than prescription medications just because they’re “natural”. Sunshine is natural, and it can give you cancer. At least prescriptions are regulated and studied rather than people thinking they can use as much as they want of a “natural product” just because it’s “natural”.
Again, I digress. Sorry. Putting that soapbox away now. (Image)
Vick’s VapoRub contains three main active ingredients: camphor, menthol, and eucalyptus oil. Let’s look at each of these components a little more.
Camphor is a bicyclic monoterpene ketone essential oil made from the wood from the Cinnamomum camphora tree. It is also found in several other species of plants, including lavender, rosemary, and basil. Historically, it has been utilized for its pungent aroma, including being used to fumigate rooms during the Black Death of 14th century Europe and to perfume bodies in preparation for burial. But it’s also recognized as so much more than a perfume.
Camphor has demonstrated antimicrobial, anti-inflammatory, analgesic, antioxidant, and neuroprotective properties. It repels pests and is used in plastic production. Pharmacologically, it modulates transient receptor potential (TRP) channels, which is thought to produce a counterirritant - or distraction - effect so that pain is lessened. (Interestingly, this is similar to how capsaicin works as an analgesic!) As an antitussive, it is thought to stimulate TRP cold receptors in respiratory passages, producing a cooling effect that decreases the urge to cough.
Menthol is a terpene alcohol derived from the mint family of plants, including peppermint (aka Mentha piperita). Similar to camphor, it is thought to contribute to antitussive effects through modulation of a cold-sensitive TRP receptor pathway in respiratory passages. The feeling of “breathing easier” produced by menthol is the reason it has been included in tobacco products, in addition to masking irritation from nicotine and smoke. People would - erroneously - feel like cigarettes helped them breathe better…because of menthol!
Eucalyptus oil is an essential oil produced from the leaves of the eucalyptus genus of plants, primarily Eucalyptus globulus, although there are tons and tons of different members of this native Australian myrtle tree family used to make essential oils. Clinical effects include antibacterial, anti fungal, antiviral, anti-inflammatory, antioxidant, analgesic, and assistance with wound healing. For respiratory conditions, it falls into the categories of expectorant and anti-inflammatory, making it a theoretically useful addition to camphor and menthol.
The exact mechanism of action of eucalyptus oils is unknown. However, it is thought to act as a mucolytic and also to decrease respiratory secretions by inhibiting pro-inflammatory cytokines. It may also contribute to (again) that feeling of cold that seems to improve respiratory comfort.
Phew. Ok, so those are the components of Vicks VapoRub. Lots of distilling of plant components. So what’s the problem in kids?
All 3 of these ingredients can cause adverse effects in young children, and the FDA and AAP recommend completely avoiding products containing camphor in kids less than 2 years old. What adverse effects, you might ask?
Toxicity from rubs containing these ingredients is usually from accidental ingestion, although the oils are readily absorbed through the skin and can also lead to toxicity (more rare for this route though). Clinical signs are apparent rather rapidly (especially in the case of camphor, like 5-15 min after ingestion) and include the following:
Burning in the mouth/throat (for PO exposures)
Nausea, vomiting, abdominal pain
Dizziness, confusion, agitation, hallucinations, ataxia
Seizures
Respiratory depression
Sooo yeah. Friends don’t let friends use products with these ingredients in their young infants or toddlers. And even in older children, we shouldn’t be doing that oh-so-tempting trick of putting it in and around the nostrils for direct application. It’s a chest rub for a reason. You can’t lick your chest. You don’t pick your chest and put what you get in your mouth like kids do with their noses (refer to initial reference about snot and how these sweet babies can do gross things :)).
Also, there are now baby and child versions of Vick’s available for ages 3 months and up. These do not contain camphor or menthol (although they do still have eucalyptus, as well as rosemary and lavender). Parents should still pay close attention to the directions for application (aka if you’re putting it on the feet at night, babies should not have mouth access to those piggy wiggies!), and children should be monitored for any signs of irritation or discomfort.
What my LOTR fan brain thinks every time my child yells this and runs away.
For reference, my preschooler won’t let me get within 10 feet of him when he sees that little tub come out. He runs away yelling, “It burns!!” So even if there may be some sort of benefit from it, don’t forget to factor in actually being able to apply it. (Image)
Also, for completeness, we’ve talked about chest rubs in this section. But don’t forget that these essential oils are also used in vaporizers, inhalers, and cough drops. Check those labels and be cautious!
The tl;dr of Kids’ Cold Medicines
Now that we’ve talked about why these remedies are blacklisted in young children with colds, let’s take a quick minute to review the highlights:
Dextromethorphan should not be used for cough suppression in children under the age of 4 years. Between ages 4-6 years, it’s still not recommended first line and should only be used under the advisement of a medical provider. Honey (for ages 1+ years) is a suitable alternative.
First generation antihistamines (e.g., brompheniramine, chlorpheniramine, diphenhydramine, doxylamine) are especially risky for children under the age of 2 years, but they are not recommended for use in kids under 6 years. Look for second generation antihistamines (e.g., cetirizine) instead when trying to help runny noses.
Chest rubs containing camphor, menthol, and eucalyptus pose significant risks to children under 2 years old. If additional antitussive help is needed, try rubs without camphor or menthol, following application instructions closely, and monitor the child for irritation or discomfort. Even better, plug in the humidifier instead.