The Complete (but Practical) Guide to Aminoglycosides
A note from the tl;dr team: this article was reviewed and updated in January 2026 to ensure accuracy and relevance
Editor's Note: The ever-fabulous Stephanie Kujawski, PharmD, BCPS has graced us once again. This time, she's here to fill the void inside of you that doesn't know how to dose aminoglycosides. And she's going to do it with a style and flair all her own (read: with lots of Star Wars references).
As is the standard with Steph's posts, this aminoglycoside dosing guide is excellent. It's thorough, yet concise. Taking what could be a very difficult and "science-y" topic and breaking it down to an easy to digest page-turner. We thank Steph for writing it, and we hope you enjoy it.
To get up to speed with this kinetics/dosing series, check out Steph’s posts on Pharmacokinetics and Vancomycin.
Pharmacokinetics Dosing Wars
Episode III: Revenge of the Aminoglycosides
Hello there!
No fish were harmed during the writing of this blog
After a few months’ brain break from kinetics blogging (during which you were hopefully able to digest everything about vancomycin), I’m back to talk about the next big bear of pharmacokinetics – the aminoglycosides.
This post shouldn’t be quite as beastly as the previous Complete (but practical) Guide to Dosing Vancomycin, but I just started writing it. So maybe I shouldn’t make any promises quite yet…
There are similarities between vancomycin and aminoglycoside dosing, and we'll highlight the parallels as we go. I'll keep the format similar to my previous posts so you can easily make the comparisons.
Because of the overlapping concepts, I may refer you back to Episodes I and II if I don’t have enough energy to explain something again. Or to redraw those freakin’ awesome graphs in MS Paint again.
So, let’s get started, and we’ll see where this takes us!
Part I: Aminoglycoside Quick Facts
Before we get too far, let’s clarify what drugs fall in the aminoglycoside class. That way we’re all on the same page. When we talk about aminoglycosides, we’re referring to gentamicin, tobramycin, and amikacin.
Yes, there are some older aminoglycoside drugs (if you’ve heard of streptomycin!), but they’ve been outclassed and phased out with the "Big 3" above.
So, first point...
Aminoglycosides are bacterial protein synthesis inhibitors. And they do this by binding to the 30S and 50S subunits of bacterial ribosomes.
I know, I know, I’m taking you way back to first year biochemistry, but bear with me – I’m an MOA nerd (if you haven’t figured that out by now). Remember that ribosomes are responsible for translation of mRNA into strings of amino acids. These amino acids are used to compile proteins, which in turn are the workhorses of cell processes.
So, aminoglycosides, by inhibiting the subunits of bacterial ribosomes, prevent bacteria from producing proteins that keep them alive. Sneaky sneaky aminoglycosides…
Second point...
Aminoglycosides have concentration-dependent action. This means that a particular concentration of antibiotic has to be achieved in order to knock off the bacteria, but it really doesn’t matter how long that concentration is maintained.
For more on concentration-dependent action, check out this guide to antibiotics.
Third point...
Aminoglycosides exhibit linear kinetics…just like vancomycin! Recall that this means a change in dose will produce a proportional change in serum concentration. It also means that a constant proportion of drug is eliminated over time.
*Refer back to Episode I for a more detailed explanation of concentration versus time-dependent antimicrobials as well as linear versus nonlinear kinetics.
Last point...
2023 was a big year for aminoglycosides (kind of in a bad way?). For starters, the Clinical and Laboratory Standards Institute (CLSI) updated the aminoglycoside minimum inhibitory concentration (MIC) breakpoints. Some key updates include lowered breakpoints for gentamicin, tobramycin, and amikacin susceptible breakpoints for Enterobacterales. What exactly does this mean? It means that the cutoff for calling an organism “susceptible” is now stricter. While aminoglycosides were generally great for treating gram-negative infections, they now have “weaker” coverage against Enterobacterales species such as Klebsiella, Citrobacter, Salmonella, E. coli, Proteus, Shigella, and Serratia spp.
Second, gentamicin breakpoints were removed for Pseudomonas aeruginosa, tobramycin breakpoints lowered, and amikacin limited to urinary isolates. Again, what does this mean? It means that gentamicin should no longer be used to treat Pseudomonas spp., amikacin can only be used to treat Pseudomonas in the urine, and tobramycin can be used for systemic Pseudomonas infections but just be aware that there is increased resistance compared to the past.
All that being said, no changes were made to aminoglycoside coverage of gram-positive organisms. Using aminoglycosides in combination with a cell-wall active agent (like beta-lactams) for synergy is still recommended. Think of this strategy as the beta lactam opening the cell wall door for the aminoglycoside to go in and fulfill its death mission from within the bacteria itself.
I know. I threw a lot of information at you. Here’s an easier to read summary version of pretty much everything I just said:
Empiric or definitive treatment of gram-negative infection (not Pseudomonas spp.) —> use tobramycin (preferred), amikacin (alternative), or gentamicin (alternative)
Empiric or definitive treatment of systemic Pseudomonas infection —> use tobramycin
Empiric or definitive treatment of Pseudomonas urinary tract infection —> use tobramycin (preferred), or amikacin (alternative)
Gram-Positive synergy —> use gentamicin (preferred), amikacin (alternative), or tobramycin (alternative)
Part II: Aminoglycosides and Concentrations
Just like with vancomycin, we are targeting particular serum levels when we use any of the aminoglycosides. However, unlike vancomycin, we actually care about both peak levels AND trough levels.
Think about the peak levels of aminoglycosides as a measure of efficacy. Remember, they're concentration-dependent drugs, so we need to reach a certain concentration in the serum in order to stop those nasty bacteria.
Conversely, think about the trough levels of aminoglycosides as being correlated with toxicity or safety. If we don’t let the drug wash down to appropriate levels between doses, then we put patients at risk for adverse effects – namely nephrotoxicity and ototoxicity!
Typically, aminoglycoside-induced nephrotoxicity is considered reversible. We use hydration, we alter the dosing (ideally switching to other medications if possible), and any other measures deemed necessary by nephrologists. Either way, we can usually undo any kidney damage.
On the other hand, ototoxicity is usually considered irreversible even when the aminoglycoside has been discontinued. It can consist of auditory and vestibular toxicity leading to symptoms of disequilibrium, tinnitus, and hearing loss. Risk factors include high serum concentrations, prolonged therapy, renal impairment, and concurrent/sequential use with other potentially ototoxic drugs.
Remember that price I mentioned earlier? Well there it is.
This is why it is so important to have a good understanding of how to dose and monitor aminoglycosides. And you, the pharmacist, are the expert.
Back to serum concentrations. Before we dig too deep, I do want to warn you that different institutions may recommend different target peaks and troughs. So make sure you check your specific policy first. Okay I digress. Back to serum concentrations but for real this time.
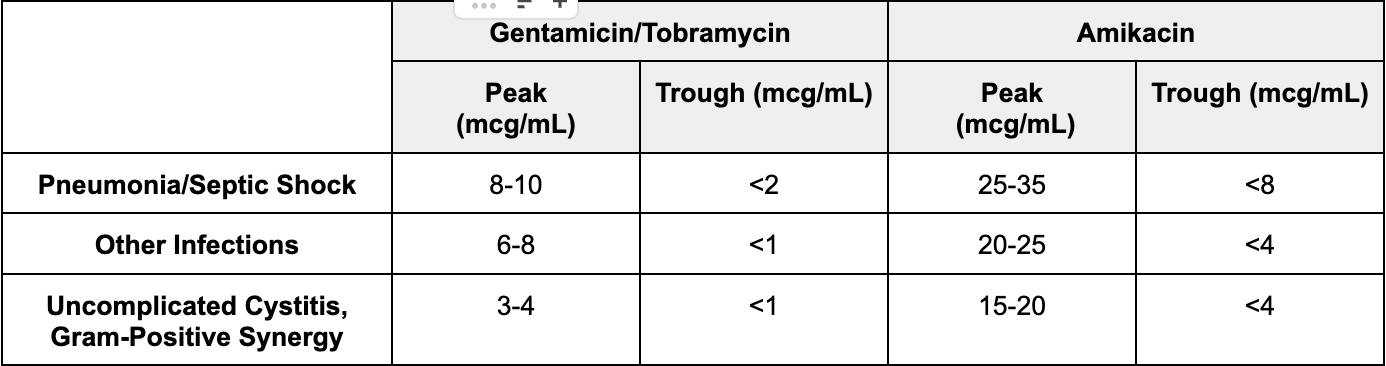
Let’s start with the easy one first: aminoglycoside troughs. Generally speaking, goal troughs are recommended to be low/undetectable in order to minimize risk of toxicity. When we’re measuring troughs, we’re not aiming for efficacy. We’re just making sure that our patient is appropriately clearing it with no signs of accumulation. In general, we aim for a goal trough of <1 mcg/mL with gentamicin/tobramycin and a trough of <4 mcg/mL with amikacin.
Okay now on to peaks. Our target peak concentration for an aminoglycoside depends on the indication. More severe infections such as septic shock may warrant higher peak goals whereas mild infections like a UTI may warrant lower peak goals. There are a lot of numbers here. Instead of writing them all out in sentences, here is a table that summarizes everything.
So here’s the real deal. For the remainder of this post, let’s focus on gentamicin and tobramycin since those are the most popular aminoglycosides.
Amikacin has its niche uses. It is sometimes still active against organisms that are resistant to gent or tobra. But its use is less common and it has different dosing targets.
I’d rather have you understand the dosing concepts of gent and tobra and be able to apply them to amikacin when needed (they're pretty much the same, you just plug in different numbers).
So from here on out, if I say “aminoglycosides”, I mean gentamicin and tobramycin, NOT amikacin.
Part III: Distribution of Aminoglycoside Doses
Let’s compare that to gentamicin and tobramycin. A "normal" adult volume of distribution for either gentamicin or tobramycin is 0.3 L/kg.
Wow. What does that tell us!?
Aminoglycosides are hydrophilic. And they don’t distribute very well throughout all tissues. They mainly stay in the extracellular fluid spaces. And they definitely don’t distribute well to cerebrospinal fluid (CSF), even in the case of significant inflammation, such as meningitis.
Now, if gent and tobra mainly stay in the extracellular fluid, what happens when this fluid compartment changes? You might get changes in aminoglycoside volume of distribution.
For a more thorough breakdown of fluid compartments, check out this post on fluid management for pharmacists.
Speaking of dosing, the low volume of distribution factor of 0.3 L/kg can help us to remember which weight to use when dosing our patients. For traditional peak and trough dosing of aminoglycosides, we use ideal body weight (IBW).
This makes sense when you think about the fact that you don’t need to account for tissue above and beyond the lean body weight since aminoglycosides don’t distribute well.
If someone is obese (aka >25% over their ideal), then we would use their adjusted body weight, calculated with a coefficient of 0.4. In other words...
Adjusted BW = IBW + 0.4 (Actual BW - IBW)
But I’m going to throw a curveball here (which I'll explain in the next section)…
Not all dosing strategies for aminoglycosides use ideal body weight! There are other dosing methods, which have been validated using total or actual body weight…
Confusing, I know.
#sigh.
Want a FREE Antibiotic Cheat Sheet?
Click the button below, and you'll get our most popular cheat sheet delivered to your inbox instantly!
Part IV: Dosing Strategies for Aminoglycosides – Traditional Dosing
The first method of dosing aminoglycosides is considered "traditional" dosing. It nearly mirrors the vancomycin dosing process, but with some slightly different numbers--so we are going to sort of breeze over it here. If you need to, review the vancomycin dosing guide for more details about the concepts.
When doing traditional dosing of aminoglycosides, we have to consider the same drug in/drug out processes that we did for vancomycin. Let's start with the "drug in" part.
Our first step is to determine the dosing weight for our patient (again, we use IBW or occasionally adjusted body weight for obese patients). Then we use population kinetic factors to calculate an estimated volume of distribution, which is generally 0.3 L/kg for gentamicin or tobramycin.
For the "drug out" portion, we are going to need to calculate a CrCl using our old frenemy Cockcroft-Gault. (If you’re wondering why the good old CG equation is a frenemy rather than a friend, remember that it's not always the best estimate for certain patients. And remember the types of adjustments we may make when calculating with this equation. And check out our Kidney Beans Part 1 post.)
Just like for vancomycin, population studies were conducted, and they determined that a population estimate for gentamicin and tobramycin could be calculated using the following equation:
Because aminoglycoside kinetics are linear, you can also calculate an estimated half-life from the equation:
So at this point, you should have the following information for traditional dosing:
- Dosing weight (ideal or adjusted, if obese)
- VD = 0.3 L/kg
- CrCl by Cockcroft-Gault
- ke = 0.0029*CrCl + 0.01
- t1/2 = 0.693/ke
And now you’re ready to start the dose finding process!
Which leads us to one of the major differences between vancomycin and aminoglycoside dosing schemes…
With vancomycin, you are mostly concerned with reaching therapeutic troughs. But aminoglycosides require attention to both peaks and troughs.
So really your first step in the dosing process is to determine your goal peaks and troughs.
Refer back to Part II above (and your drug references!) to decide what your goal peak is going to be for your patient’s particular infection. As a reminder, the target peak will depend on the indication (you shoot for higher levels for pneumonia than you do for a UTI). Goal troughs should be < 1 mcg/mL for gentamicin and tobramycin to minimize toxicity.
Then, once you’ve got your targets, it’s time to test a starting dose using steady state peak and trough calculations, just like we did with vancomycin. Traditional gentamicin and tobramycin dosing is usually:
1 - 2.5 mg/kg/dose q8 - 12h
Estimated Peak (at steady state)
Estimated Trough (at steady state)
What if your peak is not within the goal range for what you're trying to treat? Or if your trough is not < 1 mcg/mL? Then it’s back to the drawing board.
Similar principles to vancomycin apply here...
If your concentrations are only slightly off, try adjusting the dose first. Keep in mind the vial sizes (gent and tobra are both 40 mg/ml, but are available in different sizes ranging from 40 mg to 800 mg) and round to an even milligram dose.
Alternatively, if your concentrations are way off, then start with adjusting your interval. If a new interval gets you into range, great. If not, it should get you back to "slightly off" territory. Now you can make adjustments to the dose to find the sweet spot.
So there you have it. Traditional dosing of aminoglycosides. Not too bad after already plugging through the vancomycin, right?
Unfortunately, we rarely use this dosing strategy anymore.
But don't fret, that’s because there's a better way!
Part V: Dosing Strategies for Aminoglycosides – Once Daily Administration (ODA)
ODA is what we use for the vast majority of aminoglycosides today. Just a heads up, you may also see this strategy called "Extended Interval Aminoglycoside Dosing" (EIAD).
Moving on...
The really cool thing about aminoglycosides is that they exhibit what’s called a “post-antibiotic effect (PAE).” In a nutshell, PAE means that the antibiotic's inhibitory action extends beyond the period of exposure. So even AFTER the aminoglycoside concentration falls below the MIC, the bacteria are not able to grow.
Many antibiotics show some degree of PAE. But the aminoglycoside PAE is pretty much near the top of the list in terms of magnitude (along with fluoroquinolones). One might say that aminoglycosides have their very own form of The Force…
Even when you don’t think they’re around anymore, they’re still affecting their environment!
The even cooler part of this is that you, the pharmacist, get to be a Jedi!! Yes, YOU, a JEDI! You will USE THE FORCE for your own purposes – and for the greater good, of course!
We heard there was a "pharmacy to dose" order for gentamicin? Image
You are going to exploit PAE in order to maximize efficacy and minimize toxicity of the aminoglycosides.
We do this by using once daily administration (ODA). Once it was recognized that aminoglycosides have PAE, nomograms were developed that would give therapeutic peaks despite longer dosing intervals.
These longer dosing intervals would allow the drug to washout, which then minimizes toxicity. In essence, you get an "aminoglycoside-free period" in the dosing interval.
There are several validated nomograms available, but they generally operate on the same principles...
Using a specified dosing weight, you calculate a mg/kg dose
Give that dose according to a renal function-determined algorithm
Check a level some pre-determined amount of time after the dose
Based on where that level falls on your nomogram, you adjust your interval accordingly.
Seems a lot easier than the calculations in Part IV, right? Almost all of the work is done for you by the chart.
Before we take a closer look at nomograms, it is important to note that ODA is not recommended for all patients!
Here's a few of the common exclusion criteria:
CrCl less than 40 ml/min
Age less than 13 years
Pregnancy
Burns
Endocarditis (or Gram positive synergy)
Osteomyelitis
CNS infection
Surgical prophylaxis
Cystic fibrosis.
Use of the nomogram in these patients can lead to worse outcomes due to differences in distribution, metabolism, or clearance of the medication. Or in some cases, high doses are just unnecessary (e.g., surgical prophylaxis).
So, now that we’ve put that disclaimer on the table, let’s look at one of these nomograms. Below is the Hartford nomogram for once daily administration of gentamicin or tobramycin.
7 mg/kg ODA for gentamicin and tobramycin
Per Hartford, you first determine the dosing weight, which is either total body weight or adjusted (for obesity). Calculate the CrCl using Cockcroft-Gault. From there, it's a 7 mg/kg dose given according to the following renally-defined intervals:
| Estimated CrCl (ml/min) | Dosing regimen (Gent/Tobra) |
| 60 or greater | 7 mg/kg q24h |
| 40-59 | 7 mg/kg q36h |
| Less than 40 | Not recommended |
Although you can obtain your level after the first dose according to these nomograms, it’s probably best to determine if therapy is expected to continue before you go ordering levels willy nilly on everyone who gets one dose. Levels are expensive!
So, when it's time to check a level (typically within 2-3 days of starting once daily dosing), order a random serum drug level for anywhere between 6 and 14 hours after the start of the infusion.
Once you have your level, plot it on the above chart. Wherever it falls is what your dosing interval should be. Voila! You’ve just used ODA of aminoglycosides according to Hartford. How simple was that?
A few more notes about this…
If you happen to be the lucky person whose level falls ON the line, you probably want to utilize the less frequent of the 2 adjacent dosing intervals.
If your level falls ABOVE the nomogram (e.g., greater than 13 mcg/mL at 6 hours or greater than 6 mcg/mL at 14 hours), you probably need to revert back to traditional dosing since your patient’s renal clearance evidently isn’t fantastic. #KidneyProblems
If your level falls BELOW the nomogram, you may also have to convert to traditional dosing.
Something else to note is that the multiple ODA nomograms available are not all alike! Even though the procedure for using them is pretty similar, be sure you’re paying attention to how they were studied and validated.
For example, the Urban Craig nomogram is another popular option. It uses a 5 mg/kg dose (rather than Hartford’s 7 mg/kg ), but it also has curved lines that allow for a q12h dosing interval option (see below).
5 mg/kg ODA for gentamicin and tobramycin
So while those patients with spectacular renal function and aminoglycoside clearance may fall below Hartford’s lower concentration cutoff of 2 mcg/mL, the Urban Craig plot may still work because it accounts for concentrations as low as 0.5 mcg/mL!
But then you have to consider that the Urban Craig nomogram should not be used for organisms with elevated MICs >2 mcg/mL because it wasn’t validated to achieve peak:MIC ratios that high.
Usually institutions will have an established nomogram that they want their pharmacists to utilize, so it’s not like you’re going to be researching nomograms all the time and using different ones for different patients.
But long story short, pay close attention to the parameters of each nomogram because they are not clones!
NOT an image of ODA nomograms
Part VI: Save the Princess!
Meet LO. She’s a feisty 28 YOF who is 5’ 6” and weighs 61 kg. Her SCr is 0.8. Her pregnancy test is negative. She recently got herself into a bit of a pickle with some storm troopers and breathed in some nasty swamp air while hiding on Dagobah. It seems she’s developed pneumonia. The doctors would like to double cover her for Pseudomonas due to her watery exposure, and they order gentamicin (along with other antibiotics!).
Using the Hartford nomogram, what is LO’s initial gentamicin regimen?
Dosing weight for Hartford = total body weight (or adjusted if obese)
IBW = 59 kg, she is not obese
Dosing weight = 60 kg
CrCl = [(140-28)*(59)*(0.85)]/(72*0.8) = 98 ml/min
Initial dose = 7 mg/kg = 7*61 = 427 mg à 420 mg
Gentamicin comes in 40mg/mL, so an increment of 20mg is acceptable and measurable.
Initial dosing interval = q24h since CrCl > 60 ml/min
Initial regimen = 420mg q24h
LO starts her gentamicin regimen, but by day 2 of therapy, her sputum cultures aren’t very revealing. She’s running fevers as high as 102.5F, and her WBC has increased to 25. Her SCr has also increased to 1 (from 0.8). The team decides they’re going to have to do a bronchoscopy to get a better sample. What should you do?
Given therapy is going to continue based on LO’s clinical deterioration, the team’s plan to bronch, and the lack of cultures, it’s time to check a gentamicin level to make sure we’re using a good interval.
You order a 10 hour post-dose gentamicin level. It was actually drawn about 10.5 hours after the last 420mg gentamicin dose, and it returns at 6.3 mcg/mL.
What do you do with LO’s gentamicin? You plot the result!
Based on the Hartford nomogram, you would change LO’s gentamicin to a new regimen of 420mg q36h.
Continue to monitor SCr. If it continues to rise, you may need to consider traditional dosing or alternative therapies. On the other hand, if her SCr improves, you may need to adjust back to a shorter dosing interval.
Monitor her for clinical response as well, and follow up with those respiratory cultures to de-escalate therapy as soon as possible.
Basically, consider drawing another level whenever there is a significant change in labs or clinical status.
That’s it, you saved the princess! Soon enough, she’ll be back to her old adventurous self.
Well this concludes Episode III: Revenge of the Aminoglycosides. I hope this helps with your dosing adventures!
Stay tuned (and buckle up) for Episode IV: Phenytoin Wars, now available to you!