Hypertension: A tl;dr pharmacy overview
This article is under construction. Please see the 2025 AHA/ACC Guideline for the Prevention, Detection, Evaluation, and Management of High Blood Pressure in Adults for the most up-to-date information.
Courtney’s Note: Welcome back to yet another long-overdue article update! Hypertension is the most prevalent modifiable CVD risk factor and is the leading cause of death and disability worldwide, so being well-versed in it is super important. Lucky for us, the new hypertension guidelines were released earlier this year! This article was originally written by our very own Brandon Dyson, so you know it’s going to be a good one. Let’s get into it!
Hypertension (HTN), or high blood pressure, has been called the “silent killer”. It’s usually asymptomatic, unless your blood pressure is high enough to classify as a "hypertensive urgency/emergency” (only then do you really notice anything). You'll get headaches, dizziness, blurry vision, shortness of breath... that sort of thing.
Otherwise, a hypertensive patient usually feels normal. But behind the scenes, bad stuff is going down in the body.
The other silent killer. (Source)
Having high blood pressure means that your heart has to work harder. It has to overcome the added resistance it is facing with every single beat. The problems you see with HTN don’t show up in a few months. Rather, they take years to develop. But the problems are pretty serious...
Heart disease. Strokes. Kidney failure.
But most of us tend not to think of our “Future Selves” when dealing with our health. We feel fine right now, so we assume nothing is wrong. Why is this a problem?
Because it leads to HTN medications having piss poor compliance. Patients will go months without refilling them, and they see no problem with that.
You can kind of understand their point of view. If you don't feel any better (or even different) from taking a drug, why keep taking it? Why spend money on it? You may even feel worse due to nausea or hate having to pee all the time (if taking a diuretic).
The not-so-popular version of Oregon Trail.
Antihypertensives are not like pain medications or antihistamines where you can feel an noticeable difference quickly.
This makes HTN an area of impact for pharmacists. We can boost patient compliance by educating about long-term consequences of HTN. And by finding ways to minimize the cost or convenience of the patient's regimen.
You won't make a difference with every patient. But if you have an impact on even a percentage of your patients, that's better than nothing.
Diagnosis of Hypertension
Being that we are future/current pharmacists here, we're going to focus primarily on the drugs used to treat HTN. We won’t go over too many of the specifics for diagnosis. To fully whet your diagnostic appetite, please check out the 2025 AHA/ACC HTN Guidelines (technically they are the AHA/ACC/AANP/AAPA/ABC/ACCP/ACPM/AGS/AMA/ASPC/NMA/PCNA/SGIM Guidelines, but that’s a bit of a mouthful).
That said, we will at least cover the basics.
When looking at the diagnosis of HTN, there are two main branches:
Primary (previously “Essential”) HTN: The root cause is unknown. In general, lifestyle and genetics are thought to be contributing factors. But really, we all know it's mostly lifestyle, right?
Secondary HTN: This is HTN caused by something else. It could be kidney disease. Or drug-induced HTN. Basically there is some identifiable cause of the high blood pressure. If we can modify/remove that cause, we can correct the HTN.
And once you've got your "branch" of HTN identified, you will usually "stage" it according to the patient's blood pressure. Staging is as follows (systolic blood pressure (SBP)/diastolic blood pressure (BP)):
Normal: <120/<80 mmHg
Elevated: 120-129/<80 mmHg
Stage I HTN: 130-139/80-89 mmHg
Stage II HTN: ≥140/≥90 mmHg
I think it’s important to note how stringent these guidelines have become in the last decade. When this article was originally published in 2016, each goal (except “normal”) was 10 to 20 points higher. For example, elevated (previously “prehypertension”) was classified as a BP of 120-139/80-89 mmHg, and stage II was ≥160/>100. As you can see, we’ve really cracked down on BP in recent years.
Alright. With all of that out of the way, let's jump to the treatments.
Treatment of Hypertension
For starters, HTN treatment should always start with lifestyle modifications. These are things that the patient can do themselves for free without introducing foreign substances in their body.
Definitely a statement, not so sure about flavor. (Source)
Lifestyle modifications include things like maintaining a healthy weight/BMI, eating a low-sodium diet (e.g. the DASH diet, which is high in fruits/vegetables and super low in sodium), regular physical activity, smoking cessation (that’s a big one!), and moderating alcohol consumption.
You know, just healthy habits that all of us should work towards. Lifestyle modifications can have a profound effect on BP (not to mention diabetes, lipids, mood/depression, heart disease… the list really goes on and on).
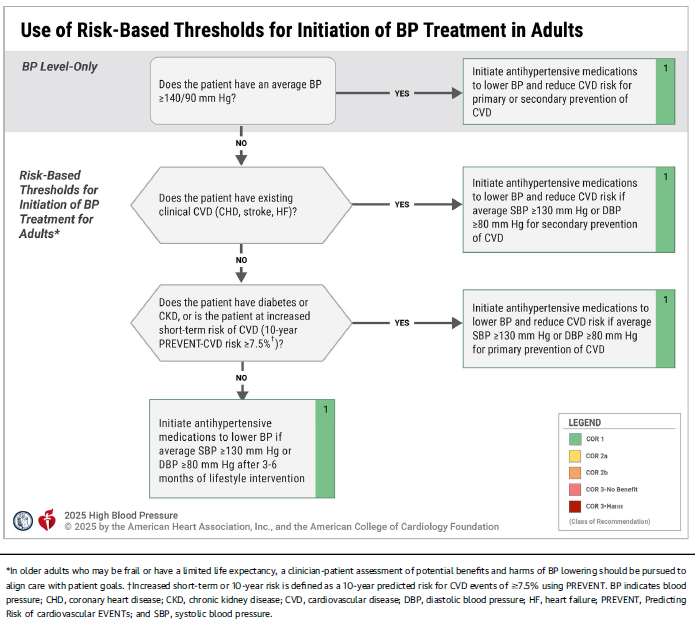
After lifestyle modifications, we can add medications. The guidelines have a handy little flowchart outlining the criteria to start pharmacologic therapy:
There are a LOT of drug classes FDA-approved for HTN. Way too many to comfortably list here. First-line options include:
Thiazide-Type Diuretics (e.g. hydrochlorothiazide (HCTZ), chlorthalidone, and indapamide)
Angiotensin-Converting Enzyme Inhibitors (ACEi) (e.g. benazepril, captopril, and lisinopril)
Angiotensin Receptor Blockers (ARB) (e.g. losartan, valsartan)
Dihydropyridine Calcium Channel Blockers (CCB) (e.g. amlodipine, nicardipine, and nifedipine)
For alternative agents, please see Table 13 in the guidelines.
Each of the four first-line classes are pretty dang good at preventing cardiovascular disease (CVD), so you really can’t go wrong with any of them. To help guide our selection of initial therapy, we look at comorbidities or “compelling indications”. For example, if our hypertensive patient also happens to be diabetic, it would be wise to start with an ACEi or an ARB to get the added benefits of renal protection.
Actually....
When you look further into it, you'll find that just about every compelling indication points to an ACEi or an ARB. Just be careful not to over-simplify and only use an ACEI or an ARB. Many compelling indications (heart failure (HF), post-myocardial infarction (MI), etc.) require multiple medications. You'll often need an ACEi/ARB, a beta-blocker (BB) and a diuretic.
Please, kids, take your medication.
In general, we can start with one drug for Stage I HTN. Once the dosage of that drug is optimized, we can add on additional agents as needed to achieve BP control. For stage II HTN, it’s recommended to start with two drugs from different classes, ideally in a single pill combination to improve adherence (because, as we talked about, adherence in this population is horrendous).
With that out of the way, let's go through our medications used to treat hypertension class by class.
Diuretics
Thiazide-type diuretics (hydrochlorothiazide (HCTZ) chlorthalidone, and indapamide) are often considered "the" first line agents for HTN.
They work in the distal convoluted tubule (DCT) of the nephron in the kidney. They inhibit the reabsorption of sodium (Na) and chloride (Cl) by blocking the Na/Cl symporter. Because water always follows sodium, this leads to the removal of fluid (i.e. diuresis).
They also are thought to decrease blood pressure due to a poorly understood chronic effect on vasodilation.
All in all, thiazides are not very effective at removing water from your patient, but they are effective at lowering BP. So that is where we primarily use them.
The primary side effects to be on the lookout for are electrolyte disturbances. They can decrease Na and potassium (K). They can actually increase calcium (Ca), blood glucose, and uric acid. And, they make you pee.
It’s also important to note that they are less effective once creatinine clearance (CrCl) dips below 30 mL/min. If you have a patient with renal disease, it may be best to try metolazone (a thiazide-like diuretic) that continues working when CrCl is less than 30.
Finally, there is a sulfa allergy risk with thiazides. The cross-sensitivity is very low. But you should keep it in mind if your patient has a history of sulfa allergy.
Contrast all that with Loop diuretics. We mentioned above that thiazides are not very good at removing fluid (despite their “diuretic” name). Loops on the other hand are incredibly good at removing fluid. They do it better than anything else. They get their name based on where they work in the kidney (by inhibiting the Na-K-2Cl symporter in the ascending loop of Henle in the nephron).
The 3 main loop diuretics are furosemide, torsemide, and bumetanide.
Because they are so good at diuresis, we don’t usually use loops for simple hypertension. We use them more for fluid build up (for example, in patients with congestive HF). And to make race horses faster.
Pee like a racehorse. Google it. (Source)
Loops lower every electrolyte…sometimes profoundly. You will need to monitor electrolytes (especially K) somewhat regularly. They can also cause acute renal failure (ARF) by dehydrating the patient. You have to keep an eye on kidney function, and you may have to temporarily hold a loop diuretic if your patient's serum creatinine (SCr) spikes.
At high doses, they can cause ototoxicity (i.e. damage to the inner ear or auditory nerve). The risk is worse if given with other ototoxic agents (like aminoglycosides and vancomycin).
There is also a sulfa allergy risk with most loops. Furosemide, bumetanide, and torsemide all have a sulfa component. Similar to thiazides, the risk of cross-reactivity is very low. But it’s worth keeping an eye on (especially on exams 👀).
If your patient needs a loop diuretic and is sensitive to the sulfa component, there is a loop diuretic called ethacrynic acid that has no sulfa reactivity (but it does have an increased risk of ototoxicity, so pick your poison I guess).
Finally, we have potassium-sparing diuretics (amiloride and triamterene). These are so named because they can retain (and slightly increase) K. They aren’t very good at lowering BP, so they are most often used in conjunction with loops and thiazides to keep the patient’s K level normal (and yes, they do help a little with diuresis/BP).
K-sparing diuretics also encompass aldosterone antagonists (spironolactone and eplerenone), which have a different mechanism of action but similar clinical effect. You will sometimes see these used in specific disease states like congestive HF or liver failure.
Your big risk with all K-sparing diuretics is hyperkalemia (i.e. high K levels). When it happens, it is usually in a patient with renal failure or in a patient on other drugs that can increase K levels.
For the aldosterone antagonists specifically, you have to worry about some weird side effects. They can cause gynecomastia (breast tissue enlargement) and erectile dysfunction in men. In women, they can reduce acne and bloating (hooray!), but at the expense of additional breast pain (boo!).
As an additional drug trivia bonus, the progesterone analog drosperinone (found in Yaz, Beyaz, and oral birth control pills) is chemically similar to spironolactone. This is one of the things that made it so popular. It reduces acne and bloating. What's not to love?
The increased risk of DVT, that's what. They can also raise serum potassium.
As you've probably noticed by now, there is rarely an "easy" answer in medicine. For every benefit obtained by a given therapy, there is an additional cost somewhere else.
RAAS Inhibitors
For those of you who played Skyrim, you should be able to read this.
Next up we have renin-angiotensin-aldosterone system (RAAS) inhibitors. The RAAS system is primarily regulated by the kidney, and it has a huge role in maintaining your blood pressure via vasoconstriction (i.e. narrowing of the blood vessels).
Angiotensin is a very potent vasoconstrictor. When it binds to your blood vessels, they clamp up like a vice, increasing your BP. Aldosterone causes your body to hold on to Na (salt) and water (water follows salt). This also works to increase your BP.
So, by inhibiting the RAAS system, we can lower BP. There are 3 types of drugs that work on RAAS:
Angiotensin-converting enzyme inhibitors (ACEi): End in –pril (captopril, benazepril, lisinopril etc). They inhibit ACE, which inhibits the conversion of angiotensin I to angiotensin II.
Angiotensin receptor blockers (ARB): End in –startan (losartan, valsartan, telmisartan, etc). They block angiotensin II from binding to its target receptor.
Direct renin inhibitors (DRI): There’s only one on the market currently — aliskiren. It directly inhibits renin (which converts angiotensinogen to angiotensin I).
The side effect profile of ACEis, ARBs, and DRIs are pretty similar. All of them can increase potassium. All of them can cause angioedema (side note: if angioedema occurs with any RAAS inhibitor, then every RAAS inhibitor is contraindicated. We do not re-challenge after angioedema because it can kill a patient pretty quickly).
They are also used in similar ways therapeutically. They’re used to lower BP. They’re used in HF. You will also see ACEis and ARBs (not so much DRIs) used for kidney protection in diabetic patients.
One word of caution on all RAAS inhibitors: They are all contraindicated in pregnancy. They are associated with fetal morbidity and mortality, so if your patient is pregnant, look elsewhere for treatment options (we’ll usually use methyldopa or labetalol in pregnancy).
A unique side effect associated with ACEis is a dry, hacking cough. This is because ACE also breaks down bradykinin in your body. By inhibiting ACE, you inhibit breakdown of bradykinin. Bradykinin accumulates, leading to a dry cough. If your patient develops a cough on an ACEi, you can switch them to an ARB or a DRI. Neither ARBs or DRIs have a direct impact on the enzyme ACE, and so bradykinin will not accumulate.
Beta-Blockers *Do we need all these extra sections??*
Like the name implies, BBs block beta-adrenergic receptors. When norepinephrine binds to a β-receptor, it activates your sympathetic (fight or flight) system. So, you can probably guess what happens. Norepinephrine leads to increased heart rate and vasoconstriction (among other things). BBs stop that from happening.
There are a few different categories of BBs. Some selectively bind to the β1 receptor, which is predominantly present on your heart (e.g. metoprolol, atenolol). Some non-selectively bind to both the B1 and B2 receptors (e.g. propranolol, nadolol). β2 receptors are primarily found in the lungs. An easy way to remember which receptor is which is that you have ONE heart and TWO lungs! Some bind to both alpha and beta receptors (e.g. labetalol, carvedilol).
And still some others are only partial agonists. These are said to have intrinsic sympathomimetic activity (ISA). Examples here are acebutolol and pindolol. The theory here is that because they only partially block the β-receptor, your heart rate and BP would be low at baseline, but could still increase if necessary (like if you were exercising, or trying to escape from a ninja).
Unfortunately, they aren’t used much clinically, because the theory didn’t work out too well in practice. They are primarily used in patients that need a BB, but are too sensitive to a “standard” BB. Personally, I have never seen one in practice… but I don’t work extensively in cardiology.
We even have some BBs that are formulated as eye drops (e.g. betaxolol and timolol). We use these in the treatment of glaucoma, because blocking the β-receptors there decrease the production of aqueous humor and decrease intraocular pressure (IOP).
Anyway, β-receptors are all over your body, and BBs are used for many different things. We use them for HTN. We give them after a patient has had a heart attack to help prevent a second heart attack. We give them in heart failure.
We even use some of the non-seletive blockers (especially propanolol) for migraine prophylaxis and as a treatment for anxiety. You'll also see them used to prevent bleeding in esophageal varicies.
You can probably predict the most common side effects of B-blockers. Bradycardia (slow heart rate) and hypotension (low blood pressure). Also, you must titrate a patient off of the drug if you are going to discontinue it. Otherwise, you could precipitate a hypertensive emergency and possibly a heart attack or stroke.
Calcium Channel Blockers (CCBs)
There are 2 classes of CCBs, the dihydropyridines (DHPs) and the non-dihydropyridines (non-DHPs).
The DHPs are a little more selective for blood vessels (compared to the heart), so they are used mostly to treat blood pressure. These all end in “-pine.” Amlodipine, nifedipine, nicardipine, felodipine and so on.
The non-DHPs are more selective for the heart (compared to blood vessels). We use these primarily to treat heart arrhythmias (but they are sometimes used for blood pressure).
There are only two non-DHPs, diltiazem and verapamil.
So even though both classes of CCBs block calcium channels, they are used very differently. And they have different side effect profiles.
DHPs are pretty good at causing vasodilation. Unfortunately, this can lead to fluid accumulation and swelling (edema). Primarily, edema shows up in the ankles/feet…not too different than what many women go through in the 3rd trimester of pregnancy.
In addition, those very dilated blood vessels can cause reflex tachycardia. Your heart rate increases because it perceives a huge decrease in blood pressure. It assumes you must be dying (or being attacked by a Mega Tiger) and tries to compensate by increasing its rate.
Amlodipine is far and away the most commonly used DHP used for hypertension, because it has the least amount of side effects. Nifedipine and nicardipine are most often used in ICUs under strict monitoring because of how profound their vasodilation effect can be. There are many reports of nifedipine causing an MI (not to mention falls) because of such a fast drop in blood pressure.
Nimodipine is primarily used in specific neuro cases like sub-arachnoid hemorrhage.
For the non-DHPs, their primary side effects are decreased heart rate…because that’s what you’re using them for in the first place. However, they are both also pretty constipating, so it’s advisable to consider a stool softener like docusate when you first start a patient on diltiazem (and especially verapamil).
And because we're pharmacists, you also need to be aware of the drug interactions with non-DHPs. Both of them are moderate CYP3A4 inhibitors. And they're commonly used with drugs that go through (at least partially) the 3A4 pathway. Warfarin and simvastatin, to name a few.
Centrally Acting Agents
Next we have some medications that work centrally (from your CNS) to lower blood pressure. These are not used first line, as there are many side effects that can occur by tinkering with your brain chemistry.
In fact, sometimes they’re even used because of those side effects. You may see them used off-label to treat alcohol withdrawal or even for ADHD.
Primarily, these drugs work by decreasing sympathetic outflow (i.e. decreasing norepinephrine release from the CNS).
The most common agent, clonidine, specifically works by agonizing the presynaptic alpha-2 receptor. Stimulating this receptor leads to a negative feedback loop that causes less norepinephrine to be released (and so decreased blood pressure).
Methyldopa's active metabolite is also an alpha-2 agonist.
The other agents work via a slightly different mechanism, but the end result is the same. Less sympathetic outflow.
The most important thing you need to know about these agents is to never abruptly withdraw therapy. You must titrate downwards, just like we did with the B-blockers above. The “rebound hypertension” seen from suddenly discontinuing a centrally acting agent can be profound.
The centrally acting agents are also mild CNS depressants, and can cause sedation, dizziness, dry mouth, and even clinical depression.
The most common centrally acting agents are (in order):
Clonidine
Methyldopa
Guanabenz
Guanfacine
Direct Vasodilators
Your agent of choice here is hydralazine. But you may also occasionally see minoxidil. As the name implies, they bind directly to blood vessels (specifically arterioles) and dilate them.
Conversely, there are nitrates (things like nitroglycerine), which preferentially bind to veins and dilate them.
I won’t talk too much about the nitrates here, as they aren’t used too frequently for hypertension. Nitroglycerine is used in an acute MI (heart attack). You also can see nitrates used in heart failure (the brand Bi-Dil, which is actually a combination of a nitrate with hydralazine, is particularly effective in African American patients).
Nitrates are also used for hypertensive urgencies and emergencies.
But for the most part, they are not used in the day-to-day management of regular, vanilla flavored, hypertension.
Actually, on a similar note, minoxidil is not used often for hypertension either. It causes ridiculous vasodilation, which leads to reflex tachycardia and edema. This is similar to what we saw with the DHPs, but to a much greater degree.
In fact, minoxidil isn’t given as monotherapy. It has to be given with a loop diuretic (to remove the fluid accumulation) and with a beta blocker (to stop the reflex tachycardia).
Yes, that minoxidil. (Source)
Also minoxidil causes hypertrichosis…or hair growth. This is why you will find a topical preparation of it over the counter at your local CVS or Walgreens. It’s called Rogaine.
All of that out of the way...
Hydralazine is somewhat regularly used for blood pressure. It’s not a first line therapy, but it can be effective as an add on. It also is a good choice for hypertensive urgencies/emergencies because it works pretty quickly as a direct vasodilator (it actually stimulates the release of your body’s own nitric oxide to cause vasodilation).
The big “NAPLEX-worthy” test question you will encounter with hydralazine is that it can cause drug induced lupus (DIL). This is somewhat similar to the systemic form of lupus (systemic lupus erythematous, SLE).
It’s an auto-immune reaction which can cause fever, muscle aches, and fatigue. The tell-tale sign of hydralazine’s DIL is a “butterfly rash” (malar rash) that goes under the eyes and across the bridge of the nose.
You can google image butterfly rash to get a visual if you like.
It appears to be somewhat dependent on dose and duration of hydralazine therapy. But there is no cut and dry dose-response curve for DIL.
Luckily, the reaction will go away if you discontinue hydralazine.
Alpha Blockers
Last, and…well, yeah, actually least…are the alpha blockers. These were originally formulated for hypertension. They block the alpha 1 receptor (predominantly on blood vessels), and block the signals for vasoconstriction.
Being alpha is tough. (Source)
This causes…wait for it….vasodilation. And a drop in blood pressure.
However, they have a lot of problems. They are notorious for a “first dose effect.” This is also called orthostatic (or postural) hypotension. Basically, your body has a hard time responding to changes in blood pressure demands.
When you transition from laying down to standing up, your blood vessels need to constrict in order to fight gravity and keep the blood flowing to your brain.
Alpha blockers stop that from happening.
And can cause a lot of blacking out (and subsequent falls).
So it’s advisable to tell patients to take these before bedtime, so they will have a few hours to let drug run through it's peak serum concentration. You also must advise them to take it slow when transition from sitting to standing. At least for the first couple of weeks.
Now a days, alpha blockers are primarily used to reduce the symptoms of BPH (an enlarged prostate). We preferentially use highly specific ones that don’t have much effect on blood pressure. You'll most commonly see tamsulosin (which has the descriptive brand name of Flomax) here.
However, for some patients that have BPH and hypertension, we may try one of the original alpha blockers. That way we can improve our BPH and our hypertension with one drug.
The three agents you’ll see here all end in –zosin.
Doxazosin
Terazosin
Prazosin
Combinations
This post is already long enough (over 3000 words) without going into each and every combination. Just know that it is sometimes necessary to put patients on two or more antihypertensive agents.
And many of these combinations already exist in a single tablet.
It will be important for your NAPLEX to know the brand names (and individual components) of these combinations.
But for now, this should be enough clinical pearls on hypertension to whet your appetite for a while.