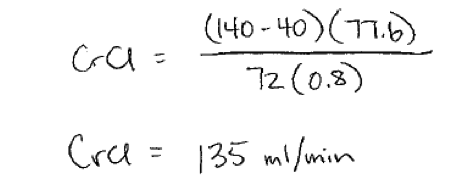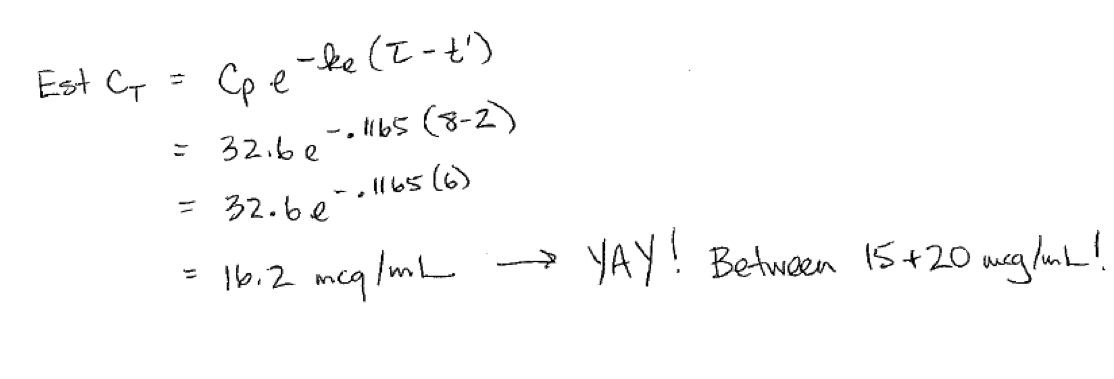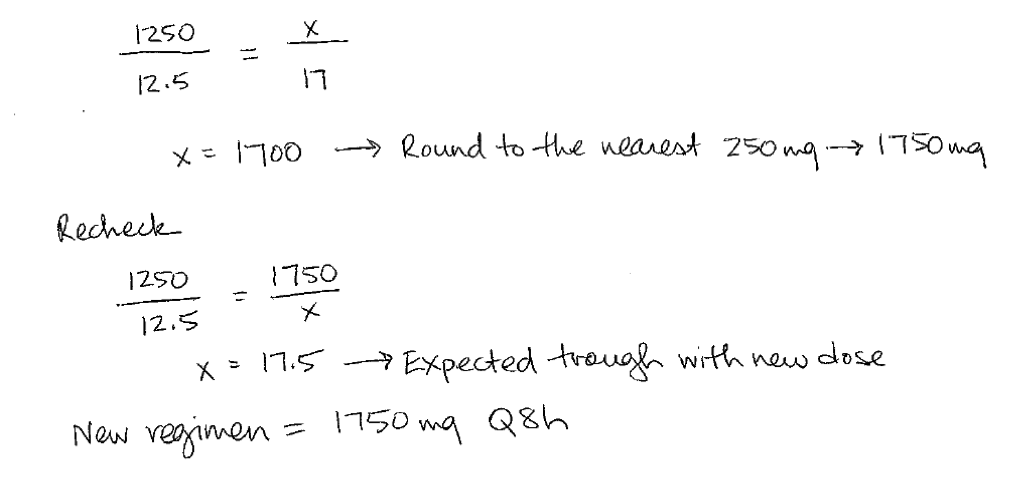The Complete (but Practical) Guide to Vancomycin Dosing
Editor's Note: She's baaaaaaacccckkk... Stephanie Kujawski, PharmD, BCPS is back with the next installment in her epic series: Pharmacokinetics Dosing Wars. Up for today, we have Episode II: Attack of the Vancomycin. It seems that our hero, Han Solo, has contracted a nasty MRSA infection (which apparently you can do while being frozen in carbonite).
Kidding aside, this post is massive (it weighs in at just over 4,200 words). But if a better guide for vancomycin dosing exists, then I am unaware of it. It's really good. Bookmark this page and spend a few days here if you need to. By the end of it, you'll be an expert on vancomycin. Take it away, Steph!
Pharmacokinetics Dosing Wars
Episode II: Attack of the Vancomycin
This poor Wampa just lost his arm. Whatever shall we treat his pending MRSA infection with?
Hello, nice to see you all again! I guess you’re all gluttons for punishment for coming back for another round of kinetics – or perhaps you’re still just staring deer-in-the-headlights at your kinetics professor! Either way, glad to see Episode I didn’t scare you off too badly, and perchance it even helped a little.
So today we’re going to tackle vancomycin. Or at least attempt to. It’s a BEAST, more beastly than a Wampa!
So this is a long one. Don’t say I didn’t warn you....
Part I: Vancomycin versus Bacteria
First let’s talk a little about how vancomycin kills bacteria. In terms of very broad categories, vancomycin affects cell wall synthesis.
Now, sure, there are many other antibiotics that also affect cell wall synthesis, including the beta lactams. But vancomycin is a little different. It binds to the D-alanyl-D-alanine subunits of the bacterial cell wall structure, which prevents peptidoglycan polymerase and transpeptidation reactions.
Just for comparison, remember that beta lactams bind directly to transpeptidase (a bacterial enzyme that cross-links the cell wall). So both vancomycin and beta lactams prevent cross-linking in the bacterial cell wall. They just do it at different steps of the pathway.
In normal terms. Think of the bacterial cell wall as a house being constructed. If you've ever seen a building project in process, you've seen what looks like a spider web of 2x4s "framing" out the house. These 2x4s are cross-linked to provide stability to the house. Cross-linking serves the same purpose in a bacterial cell wall.
And vancomycin binds to the structure and doesn’t let the bacterial enzymes do their cross-linking construction jobs.
Because the cell wall is then not fully functional, the cell (aka the bacteria) ends up also not functioning. In most cases, vancomycin actually KILLS the bacteria; therefore, it is deemed bacteriocidal. However, in some less common cases, vancomycin only stunts the growth of bacteria, making it bacteriostatic.
We should also pause here and make sure one thing is clear…vancomycin inhibits cell wall synthesis....and it is ONLY active against Gram POSITIVE bacteria!
Those sneaky Gram negatives still have a cell wall. It's just thin and wimpy and sandwiched between two cell membranes. Also, Gram negatives don't have the D-ala-D-ala sequence in their cell walls. So vancomycin is utterly useless against them.
However, Gram negatives still have the transpeptidase enzyme, so beta lactams are still effective.
It should also be noted that even some Gram positive bacteria are sneaky and have come up with ways to resist vancomycin’s actions, such as using D-lactate in their cell walls instead of D-alanine, which prevents vancomycin from binding.
For more info, refer to this awesome primer on antibiotics.
Part II: Vancomycin and Concentrations
Alright, now that we have that vancomycin background, we can move onto kinetics. Based on our discussion of antibiotic kinetics in Episode I, we know that most antibiotics act in either a time-dependent or concentration-dependent manner.
But I’m going to throw a curveball here.
Although vancomycin historically has been classified as a concentration-dependent antibiotic, in more recent years, it has been informally classified as “exposure” or “AUC-dependent”. What this means is that its killing action is really dependent upon both the concentrations reached in the body as well as the time that those concentrations are maintained. It's kind of like a daywalker. It's both time and concentration-dependent.
So, if you think of your time-concentration curves, exposure is graphically represented by the AUC.
All of the drawings on this post are original artwork by Stephanie Kujawski, PharmD, BCPS. Please contact Stephanie if you are inspired and would like reprints to hang in your house.
So as pharmacists we must pay close attention to the concentrations we are achieving in the body in order to ensure we are adequately treating an infection. These days, with resistance rates what they are, we usually want to target trough levels of 15-20 mcg/mL. Sometimes we might let those trough levels range a little lower to 10-15 mcg/mL for a cellulitis indication.
If you just think "shoot for 15 mcg/ml," you'll generally be right either way. But as you learn and practice, you’ll find there are times that targeting a more specific range, like 18-20 mcg/mL for that meningitis patient, is important!
Let's take the idea of following concentrations a step further. This means we have to be able to predict how a drug enters and distributes through the body and how it leaves or is cleared from the body.
To illustrate, let's use our shopping and closet analogy from Episode I. Let’s say your roommate is fed up and has imposed a clothes limit on your exploding closet. You are only allowed to have 50-60 items in the closet at any given time. (I'm not sure how you got into such an agreement with your roommate, but you probably should have had a lawyer look at the paperwork before you signed off on it.)
Regardless, now you’re going to have to keep track of how many items you’re buying versus how many items your friends are willing to take off your hands.
As long as you keep these balanced, your closet will stay in check. But if either your buying or your friends’ willingness to take your stuff changes, then your closet may either explode again or you won’t have anything to wear!
And don't noooobody wanna see that birthday suit.
You've seen similar "in versus out" analogies in your calculus classes. Bathtubs filling with water while the drain is open. The amount of your income versus your expenses. The point is, to treat an infection we have to balance the amount of vancomycin going into the body with the amount leaving.
If we have too much vancomycin going in, toxicity will follow (won't somebody please think of the poor kidneys!?). If we have too little vancomycin going in, the infection will worsen and spread.
Part III: Distribution of Vancomycin Doses
Let’s begin by tackling the intake end of things by trying to predict how vancomycin behaves when administered to a patient.
We need to consider a few pieces when trying to predict a drug's distribution....
Is the drug highly protein bound?
Is it such a large molecule that its size may affect its ability to travel certain places in the body?
In the case of vancomycin, it has a wide range of reported protein binding - anywhere between 10-50%. It is also quite a large molecule with a molecular weight of over 1400 Da.
Just for comparison, another commonly used intravenous antibiotic, the beta lactam cefazolin, only has a molecular weight of just over 450 Da.
So you can imagine how vancomycin might get a little "distracted" on its travels through the body. It's semi-social with other proteins. And it's just too large to fit through some of the doorways in the body.
That being said, it still distributes quite well out of the blood and into tissues. This is why we can use it to treat such a variety of infections, from bacteremia to osteomyelitis. Its ability to treat meningeal infections is a little more dependent on the degree of inflammation present.
Remember that the Blood Brain Barrier (BBB) is supposed to be an impenetrable fortress that prevents things from getting into the CNS. However, with meningitis, inflammation causes the BBB to open up a bit. That inflammation is what allows a large molecule such as vancomycin to pass through and get into the CNS.
Ironic, isn’t it, that the infection itself is what allows us to use a certain antibiotic to treat it?!? As the group "Lit" once told us, "Sometimes it seems to me I am my own worst enemy..."
So, anyways, you have a patient who needs vancomycin, and you start administering doses. Each vancomycin dose goes through a process where it is administered intravenously, which causes the serum level to increase. Then the vancomycin is distributed out of the blood and into the tissues. The blood and the tissues reach a sort of equilibrium. Finally, it is eliminated from the body, which is done largely via the kidneys.
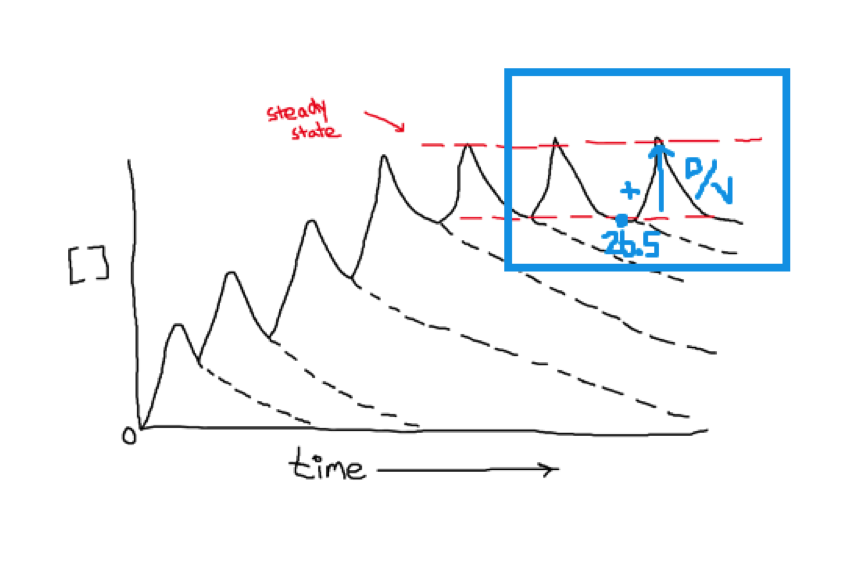
At first, the body is going to be like, whoa, what is this new stuff you’re pouring into me! So you have to fill the tank, per se. But as you start regularly administering more doses, each one is being eliminated as the next one is coming in. Eventually you reach what is called steady state.
This is the time period when serum levels are pretty constant from dose to dose, meaning each peak and trough you obtain with each individual dose should be pretty similar to the ones obtained with the last dose. Steady state is generally achieved in five half lives or, alternately, after about 4 consistent doses of a medication.
For those of you who don't remember your biochemistry classes, [ ] = concentration.
Steady state depends on a number of things. But in the case of vancomycin, because of its renal elimination, we’re looking for consistency in renal function in order to achieve this shining mecca of drug administration. When the renal function changes abruptly, we may lose steady state.
So whatever calculations we do at a particular level of renal function are only applicable if the renal function stays the same. But we’re going to try to predict drug action anyway.
In pharmacy land, we use the term volume of distribution (VD) to describe how a drug distributes through the fluid and tissues in a person’s body. We figured out the VD from population kinetic studies, in which vancomycin was administered to a bunch of people and then blood concentrations were measured and compared to the doses given. Using that info, a generally accepted VD for vancomycin is 0.7 L/kg.
And because vancomycin (somehow) distributes quite well into most tissues, we usually use a person’s total body weight to calculate this (rather than ideal or adjusted body weight). Ideal and adjusted body weight might not take into account all of a person’s muscle or adipose tissues that are exposed to vancomycin.
So in general, remember actual (aka total) body weight and a volume of distribution of 0.7 L/kg using that total body weight.
Vancomycin Volume of Distribution (VD) = 0.7 L/kg
CAVEAT #1: Some institutions practice slightly differently regarding which weight to use for obese patients requiring vancomycin. While total body weight is generally good for most patients, it might lead to problems in morbidly obese patients. So some pharmacists will use adjusted body weight in these cases.
The thought behind this is that using total body weight will eventually lead to accumulation and overshooting our target serum levels. Basically, the empty tissues soak up the vancomycin and then when they’re all good and saturated, the high doses of the drug don’t really have anywhere else to go.
They accumulate until you end up with super high trough levels and renal injury. So for people who are ~150 kg and upwards, you may want to consider using an adjusted body weight.
CAVEAT #2: For the same reasons as above, some pharmacists will also use an empirically lowered volume of distribution in their calculations for morbidly obese patients. So instead of using 0.7 L/kg, they might do 0.6 or 0.5 for people that are very large. This may help to prevent overshooting levels.
This is in no way actually a proven practice, but we’re estimating all around anyway…
So let’s practice! Meet HS. He’s a smooth 40 YOM with a penchant for flying and leather vests. He’s a trim, fit 6 feet tall, 80 kg person.
What is his vancomycin volume of distribution? Well, 0.7 L/kg * 80 kg = 56 L.
Part IV: Vancomycin's First Order Elimination
So now that we know how vancomycin distributes throughout the body, we need to examine the other end of things - how vancomycin is cleared from the body.Do you remember from Pharmacokinetics Dosing Wars: Episode I when we talked about first order kinetics? When a constant proportion of drug is eliminated over a period of time (rather than a constant amount)?
Good, because vancomycin kinetics are described by first order (or linear) processes.
Think back to your high school algebra class. You probably learned about something called decay. Described by the equation A = Pert.
You probably learned it in reference to radioactive materials (where your math teacher wanted you to find out how old some rock was by how much radioactive carbon was left in it).
It's exactly the same equation for first order drug elimination.
The A is your C2 (or for simplicity’s sake, your trough), the P is your C1 (or your peak), r is your ke (your elimination rate constant that describes how fast the drug is going away), and t is still t (the amount of time that has passed between C1 and C2).
And so, in pharmacy, first order elimination is described by the equation C2 = C1eket, where ke is intrinsically negative since it is elimination. This equation is graphically represented on our normal time-concentration curve as such:
Now let’s take just the elimination portion of that curve and take the ln (natural log) of each data point. That results in a figure that looks like this:
See how the elimination line becomes linear instead of curved?? And the slope (aka the ke) is constant?? Well that fits right in with our first order kinetics! A constant rate of elimination!
Alright, so we talked about weight and volume of distribution for the intake portion of vancomycin dosing. Now we’re going to talk about how to find the ke to help describe elimination.
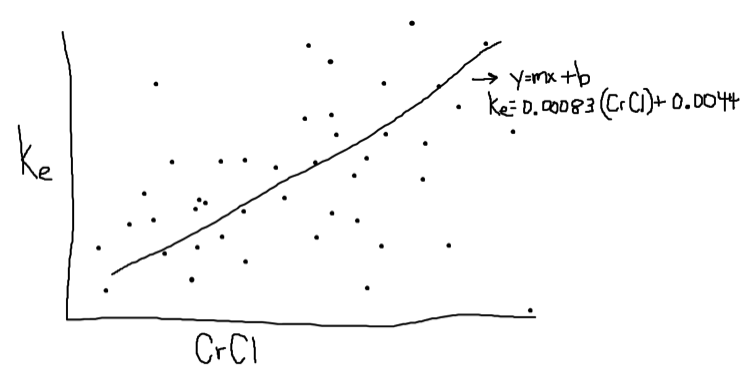
Again, based on population kinetics, it has been determined that we can estimate ke with the equation 0.00083*CrCl + 0.0044.
Vancomycin ke = 0.00083CrCl + 0.0044
Where did that come from? From the same place we estimated VD earlier. Basically, investigators again gave vancomycin to a whole bunch of people. They measured peak and trough levels. They calculated the ke from our friend C2 = C1eket. And then they plotted ke versus CrCl (calculated according to Cockcroft Gault). From this, they were able to determine the line of best fit in the format y=mx + b.
And voila, you have ke=0.00083*CrCl + 0.0044!
From the ke, it is pretty easy to find the patient’s half life. What is half life, you say? It’s the time it takes for the concentration of the drug to decrease by 50% in the body.
For first order drugs, the easy way to find half life is to divide 0.693 by ke. Graphically, half life looks like this:
CAVEAT #3: Finding k e is pretty straightforward. Plug in the CrCl and chug the math for k e . The tricky part is in finding CrCl! Of course, Cockcroft Gault is supposed to be straightforward…for more info on this lie, see our post on Kidney Beans Part 1.
Plug in the patient’s age, ideal body weight, and serum creatinine, adjust for gender, and math away!
However, it’s a little more difficult when you have a little old 95 year old lady who only weighs 45 kg and doesn't have much muscle mass. Her SCr of 0.3 and calculated CrCl of 80 ml/min are probably more fitting for her 40 year old grandson than for her.
So you may have to fudge some numbers here and there. Yes, I said you may have to fudge some numbers (#gasp!). Perhaps you round her serum creatinine to 0.8…or 0.9…or 1 for the purposes of calculation to get a more reasonable renal estimate.
Want a FREE Antibiotic Cheat Sheet?
Click the button below and you'll get our most popular cheat sheet delivered to your inbox instantly!
CAVEAT #4: In another scenario, what if you have the obese 260kg person? Are you going to use ideal body weight to calculate CrCl? Or would adjusted body weight be more appropriate since that patient has more tissue (some of which is bound to produce creatinine)??
There is no consensus on the best practice for these situations. We just know doing the black and white calculation may not be the most accurate.
So for our patient HS, if his serum creatinine is 0.8, what is his CrCl? Remember he’s a trim 80kg, 6 feet tall, 40 year old.
So, first, his ideal body weight has to be calculated:
Then, Cockcroft Gault can be employed:
Now, we have HS’s CrCl, so what is his ke based on our population estimate equation?
Ok, so at this point, we have the patient’s total body weight, as well as population estimations for volume of distribution, CrCl, and ke. It is FINALLY time to calculate a vancomycin dose.
I promised it would come, right?!
Part V: The Vancomycin Dosing Process
Some institutions believe in giving every patient a loading dose of vancomycin. The thought is that because you’re starting from scratch, you want to fill the tank to achieve target trough levels more quickly.
If you are going to load, you will usually use the patient’s actual body weight…whether or not they’re obese. Remember, you’re starting from nothing. That being said, most places cap a single vancomycin dose at either 2000 or 2500 mg.
Other institutions do not load unless a patient is septic. The thought is to prevent overshooting and causing renal injury.
You will just have to get a feel for your institution’s practices and also which patients are more critical. In general, if you are going to load, it’s a 20-25 mg/kg dose.
Regardless of loading dose, you are going to need to figure out a vancomycin maintenance dose. There are two pieces to figure out with every vancomycin regimen:
- Dose
- Interval
It's a bit of "guess and check" to determine how a given dose is going to behave in a particular patient. We figure this out by using derivations of our trusty first order equation.
We need to estimate what a particular regimen’s peak is going to be at steady state so that then we can calculate an estimated trough at steady state…
Which hopefully will be between our goal of 15-20 mcg/mL.
So first, let’s estimate the dose we want to give. In general, a good place to start is 15-20 mg/kg. Then round your dose to the nearest 250mg (technicians won’t particularly like having to draw up a dose of 1289 mg…just go ahead and make that 1250mg).
Next, let’s determine an interval to give this dose. Here are some general rules to start with:
- q8h for CrCl > 100
- q12h for CrCl 60-100
- q24h for CrCl < 60
- (Hemodialysis is another beast that we won’t address here...you’re probably already bleary eyed.)
So for HS, we get a range of doses of 1200-1600 mg. Let’s choose 1250mg to start. Based on his CrCl, we would choose an initial dosing interval of q8h.
Now that we have our dose and interval, we need to see what it’s going to do in our particular patient. It’s time to plug and chug our data into the estimated steady state peak equation. We need the dose, infusion time (usually no faster than 15 mg/min), ke, volume of distribution, and interval.
Of note....
Even though there’s no “target” peak, per se, vancomycin toxicity is associated with supratherapeutic troughs. We usually shoot for peaks < 45 mcg/mL. Any higher and you’re not going to get the troughs you want.
Now that we have our estimated steady state peak, let’s get the estimated steady state trough using our equation.
If your trough is not within the desired range of 15-20 mcg/mL, you have a decision to make. Do you change the dose? Or do you change the interval?
A good rule of thumb is that if you are just slightly off from the goal trough level, try changing your dose first. If you are drastically off (i.e. estimating troughs of 27…33 mcg/mL), you’re going to have to change the interval. You won't be able to change the dose enough to get that into range!
Once you have a regimen that is meeting your desired trough goals, it’s time to move forward! If you didn’t load your patient, go ahead and recommend/enter your maintenance dose.
If you did happen to give a load, you need to determine when to start your maintenance dose after the load finishes.
This can be done using our trusty first order equation.
C2 = C1eket
C2 is the serum concentration at which we want to redose the patient. In most cases, it's going to be 15 mcg/mL. C1 is the peak concentration obtained from the load (which is the loading dose divided by the estimated volume of distribution (LD / VD) since we’re starting giving the load to an empty tank).
You have an estimated ke, and you need to solve for t. Remember, we’re trying to find how long after the loading dose FINISHES we have to wait to give the first maintenance dose (so the count starts at the end of the infusion).
Solve for t and voila, you have your time to wait! Enter your dose, and you’re golden!
Part VI: Vancomycin Levels and Adjustments
After you get your dose going, it’s up to the medical team’s and your discretion as to if and when to check a vancomycin level. If the patient is clinically improving and their renal function is stable, you might get away without checking one. If vancomycin isn’t expected to continue very long (greater than a few days), maybe you don’t need to spend the money on the level.
IF, however, the patient is not improving, their renal function tanks, or they’re going to be on an extended course, (especially if it’s the little old lady or the morbidly obese patient from earlier), perhaps THEN you want to check a level.
If you do check a level, try to get one at steady state. Troughs should be drawn about 30 minutes prior to the next dose.
Quick side note. ALWAYS make sure the trough was drawn at the correct time before reacting to it. It's very common to see low or high troughs just because they were drawn inappropriately. The first question to ask when looking at a trough is, "Was this drawn at the right time?"
If your trough is only slightly off from goal range, no problem. Remember vancomycin has LINEAR kinetics! Changes in dose should produce proportional changes in serum level, as long as the dosing interval remains constant.
So a simple proportion is often all that is required to find your new regimen.
Let’s pretend HS’s appropriately timed vancomycin steady state trough on 1250mg q8h returned at 12.5 mcg/mL. And let's say you’re trying to keep his troughs between 15 and 20 mcg/mL. Since it’s just a little off, let’s do the proportion method:
The trickier case is when your trough level is way off goal. Just changing the dose may not be adequate to achieve the trough you want. So you’re going to have to change the interval…which means cycling back through the guess and check calculations.
The advantage of having a patient specific drug level is that then you can calculate a patient specific ke to use in the equations (rather than just a population based ke from the equation).
We get to use the same trusty equation (see a pattern???).
C2 = C1eket
Your C2 is the trough you obtained. The C1 is your peak steady state concentration (which can roughly be calculated by dose divided by volume of distribution, plus your trough!)
D/VD + Trough = C1
It’s the same idea as the estimation of your loading dose peak, except now you’re not starting from scratch. You’re starting from your steady state trough level.
You know t to be the time between peak and trough on the same dosing interval. Then solve for ke. (And sneaky tip…now you can back calculate an estimate of CrCl using your population kinetics ke equation. It's not 100% accurate, but it's a more patient-specific estimate). (see below figure)
So let’s say HS’s steady state trough from a regimen of 1250mg q8h returned at 26.5 mcg/mL (#eek!). His last dose prior to the level was infused over 2 hours starting at 1200. And the level was drawn appropriately at 1930.
What is HS’s ke? What should his new dose be?
Let’s start by representing the problem graphically.
This is a figure that shows one dose of the vancomycin. Specifically the dose right before the level was drawn. We see the start of the infusion at 1200 (yes, of course we know that, since we are at steady state, the starting concentration isn’t zero, but bear with my chart drawing skills).
The dose infuses over 2 hours until 1400, at which point we know there’s a C1 peak. But we don’t know what that is since we don’t actually check vancomycin peaks. Then, we know there are 5.5 h in between the end of the infusion and when the level was drawn.
And we have our unknown ke....
So essentially we have an equation at this point with 2 unknowns, C1 and ke, where, remember ke is inherently negative since it’s elimination:
Now, you can’t find ke at this point…but you can get rid of one unknown by estimating C1.
Remember when we were trying to figure out how long after our loading dose to wait to give the first maintenance dose? Remember we said concentration is dose divided by volume to get our initial C1 for the load?
Well this is a similar situation, except instead of starting from scratch as we did for the load, we’re at steady state with a trough of 26.5. So our steady state peak concentration should be our dose divided by volume of distribution added to our trough level.
Cpeak(ss) = Dose/VD + Trough
Visually:
So, once we find C1, we’re only left with ke as an unknown in our trusty equation. And we can solve for a patient-specific (rather than a population estimate) ke! See here:
Yay! So, now that we have HS’s specific ke, sometimes it’s handy to back calculate a CrCl using our original ke = 0.00083 * CrCl + 0.0044 equation.
Yes, I know we’re using a population equation to back calculate a patient-specific estimate, but this is still an estimate more specific to your patient than Cockcroft Gault.
If we do this for HS, we get an estimated CrCl of 128 ml/min, which isn’t too far off from our earlier estimate of 135 ml/min. However, for some reason he doesn’t seem to be clearing the vancomycin like we thought he would. We know this because of his supratherapeutic trough!
But now, armed with his specific ke, we can decide on a new dosing regimen. Using our steady state estimation equations we can get back within our goal trough range of 15-20 mcg/mL.
This is the same guess and check process we used with the initial regimen, just now with a patient-specific ke.
So once you come up with a reasonable new regimen that meets our goal level criteria, you have to decide how long you are going to wait before starting said regimen.
We know we need to let the patient’s blood levels wash down a little bit since his trough is 26.5. But the question is how long?
Well, guess what (spoiler alert)? We’re going to use our same trusty equation to find this out.
C2 = C1eket (you will learn this equation by the time I'm through with you ;) )
Just like our load, we want to wait until the serum level washes down to about 15 mcg/mL (C2) from the current level of 26.5 mcg/mL (C1). We know our ke for our patient (0.111/h), and we need to solve for t.
Once you have t, you know when to resume therapy with your new maintenance dose!
Part VII: Just Kidding
Ok, breathe!!! In a nutshell, that is how we dose, monitor, and adjust vancomycin therapy. Take a moment to sit back and consider all of the estimations...on estimations...on estimations. It's crazy, isn't it?! Hence, the importance of knowing when to check serum levels to get a true answer.On that note, happy trails practicing your vancomycin! It takes time, but eventually you’ll see all the ways that the one trusty equation is the Chewbacca to your Han Solo. You'll also learn how to navigate the gray areas of obesity, renal dysfunction, and when to check levels, but never hesitate to bounce your plan off of someone with more experience.
Remember: C2 = C1eket
If you made it through this entire post, you legitimately deserve a medal. No, but really. I’ll recommend you to the Resistance for recognition by Leia.
And FYI, Episode III: Revenge of the Aminoglycosides is available for more kinetics fun!



![For those of you who don't remember your biochemistry classes, [ ] = concentration.](https://images.squarespace-cdn.com/content/v1/54fcabf1e4b05f6987ac7401/1464263884188-16IAF68LY2NJR8N2GBVV/image-asset.png)